https://doi.org/doi:10.64898/2026.04.20.719588
https://pubmed.ncbi.nlm.nih.gov/42079090/
#Drosophila #Metabolism
Job Alert
Applications for PhD Scholarships, Postdoctoral Fellowships & Visiting Researcher Grants
Location: Danmark, Odense
Deadline: 2026-08-18
#hiring #PhD #Postdoc #Endocrinology #DiabetesResearch #LifeSciences #associateprofessor #metabolism #medicine
Metabolic effects and adrenergic upregulation in hyperthyroidism USMLE
#USMLEStep1 #MedEd #Endocrinology #Physiology #Hyperthyroidism #Thyrotoxicosis #Metabolism #Step1Prep #HighYield #MedSchool #InternalMedicine #ThyroidHealth #T3 #T4 #FutureDoctor #StudyGram #MedTwitter #Anki #BoardExams #Medical
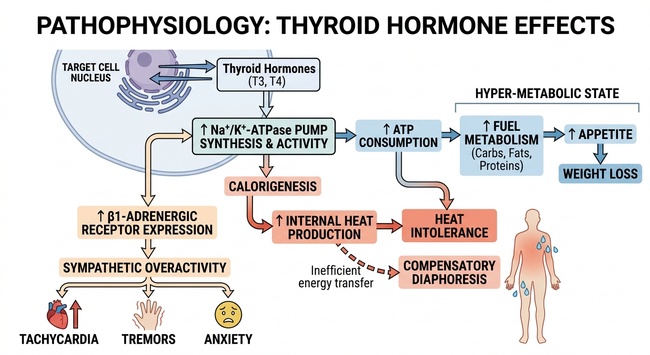
Master the metabolic impact of Hyperthyroidism (U01.17.087) for USMLE Step 1. Understand how excess thyroid hormone increases BMR, upregulates $\beta$-1 receptors, and drives heat intolerance through uncoupling protein (UCP) expression. High-yield guide on mymedschool.org.
An anaerobic pathogen rewires host #metabolism to fuel oxidative growth in the inflamed gut.
ETBF uses its toxin to reshape epithelial metabolism, creating a local oxidative niche that fuels its gut colonization.