In a revolutionary breakthrough, scientists have discovered a way to guide stem cells to develop into fully formed organs, opening new frontiers in disease treatment and tissue repair. Stem cells, known for their incredible ability to become any cell type in the body, can now be directed with precision to form complex organ structures.
This discovery could transform medicine, offering hope for patients with damaged organs, chronic diseases, or injuries that currently have limited treatment options. Researchers are exploring how this technology can create functional organs in the lab, potentially reducing the need for donor transplants and eliminating long waiting lists. By understanding the signals that tell stem cells how to grow, scientists can replicate the natural process of organ development in controlled conditions.
This breakthrough not only promises life-saving therapies but also paves the way for personalised medicine, where organs can be grown using a patient’s own cells, dramatically lowering the risk of rejection. As research progresses, the ability to guide stem cells may revolutionise how we approach healthcare, making organ failure a challenge of the past. The potential to heal the human body from within has never been closer to reality.
General science & breakthrough
#ScientificBreakthrough #MedicalRevolution #FutureOfMedicine #NextGenHealthcare #LifeSciences #RegenerativeMedicine
Stem-cell and organ-growth focus
#StemCells #OrganEngineering #LabGrownOrgans #BioPrinting #TissueRegeneration #CellTherapy #Organogenesis #PersonalisedMedicine
Impact & hope
#EndOrganShortage #HealingFromWithin #HopeForPatients #MedicineOfTomorrow #ChronicDiseaseCare #TransplantAlternatives
ReBone is a four-year Marie Skłodowska-Curie Doctoral Network funded by Horizon Europe.
Our mission? To train a new generation of researchers to design personalized bone-substitute implants using bioactive glass/ceramic materials — combining biomechanics, materials science, clinical research, and advanced manufacturing.
We're tackling urgent challenges in bone repair:
🔹 Patient-specific solutions
🔹 Better mechanical reliability and bioactivity
🔹 Optimized manufacturing processes
—all with the goal of improving outcomes for people with critical-size bone defects.
🌐 Want to learn more?
Check out our brand-new website:
👉 https://rebone.eu
You’ll find info about the project, partners, DCs, research progress, training activities, and events.
📰 And yes — Issue 1 of the ReBone Newsletter is out now!
Get a snapshot of our latest research, events, and what’s ahead.
📥 Read it here: https://rebone.chem.polimi.it/s/6TdW92mfRTCmC27
#ReBoneProject #MSCA #HorizonEurope #BoneImplants #BioactiveCeramics #Biomechanics #TissueEngineering #PhDLife #OpenScience #PersonalisedMedicine #ResearchCommunity
#AnyaPlutynski - #Philosophy of #MolecularBiology & #Genes
https://www.youtube.com/watch?v=lKhaVhoAS5o
#PhilosophyOfBiology #Biology #PhilosophyOfScience #Science #CancerGenomeProject #Cancer #Genome #Disease #Mutation #Medicine #Epigenetics #PersonalisedMedicine #CloserToTruth #RobertKuhn
Anya Plutynski - Philosophy of Molecular Biology & Genes
Qianhan Liu et al. explore the power of machine learning in #radiomics to predict T2/T3 staging in laryngeal and hypopharyngeal cancer - a promising step towards precision medicine in oncology.
#EuropeanRadiology #PersonalisedMedicine #CancerResearch
🔗 https://link.springer.com/article/10.1007/s00330-023-10557-8
Machine learning model to preoperatively predict T2/T3 staging of laryngeal and hypopharyngeal cancer based on the CT radiomic signature - European Radiology
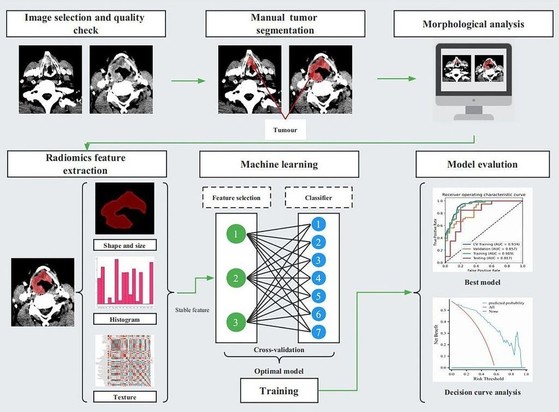
Objectives To develop and assess a radiomics-based prediction model for distinguishing T2/T3 staging of laryngeal and hypopharyngeal squamous cell carcinoma (LHSCC) Methods A total of 118 patients with pathologically proven LHSCC were enrolled in this retrospective study. We performed feature processing based on 851 radiomic features derived from contrast-enhanced CT images and established multiple radiomic models by combining three feature selection methods and seven machine learning classifiers. The area under the receiver operating characteristic curve (AUC), accuracy, sensitivity, and specificity were used to assess the performance of the models. The radiomic signature obtained from the optimal model and statistically significant morphological image characteristics were incorporated into the predictive nomogram. The performance of the nomogram was assessed by calibration curve and decision curve analysis. Results Using analysis of variance (ANOVA) feature selection and logistic regression (LR) classifier produced the best model. The AUCs of the training, validation, and test sets were 0.919, 0.857, and 0.817, respectively. A nomogram based on the model integrating the radiomic signature and a morphological imaging characteristic (suspicious thyroid cartilage invasion) exhibited C-indexes of 0.899 (95% confidence interval (CI) 0.843–0.955), fitting well in calibration curves (p > 0.05). Decision curve analysis further confirmed the clinical usefulness of the nomogram. Conclusions The nomogram based on the radiomics model derived from contrast-enhanced CT images had good diagnostic performance for distinguishing T2/T3 staging of LHSCC. Clinical relevance statement Accurate T2/T3 staging assessment of LHSCC aids in determining whether laryngectomy or laryngeal preservation therapy should be performed. The nomogram based on the radiomics model derived from contrast-enhanced CT images has the potential to predict the T2/T3 staging of LHSCC, which can provide a non-invasive and robust approach for guiding the optimization of clinical decision-making. Key Points • Combining analysis of variance with logistic regression yielded the optimal radiomic model. • A nomogram based on the CT-radiomic signature has good performance for differentiating T2 from T3 staging of laryngeal and hypopharyngeal squamous cell carcinoma. • It provides a non-invasive and robust approach for guiding the optimization of clinical decision-making.
RT by @EUScienceInnov: 🌟Proud to see #EUPartnerships steering into a future where medicine is tailored to the patient.
With #EPPerMed we aim to lead the transformative journey of #PersonalisedMedicine 🚀
Discover how #EUfunded R&I is contributing to personalised healthcare👇
🐦🔗: https://nitter.cz/IreneNorstedt/status/1722226051932356674#m
[2023-11-08 12:14 UTC]
Launch of the European Partnership for Personalised Medicine
Research and innovation news alert: The new European Partnership for Personalised Medicine will promote all areas and disciplines of precision medicine, facilitate innovation and transfer, as well as encourage its consolidation in health systems, allowing continuous improvement.
#IDMastodon #infectiousdiseases #immunology #ARDS #Sepsis
👏 @EliP
#IDMastodon #infectiousdiseases #microbiology
@infectiousdiseases @microbiology @MicrobioJC #PersonalizedMedicine #personalisedmedicine
https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciad467/7240295?searchresult=1
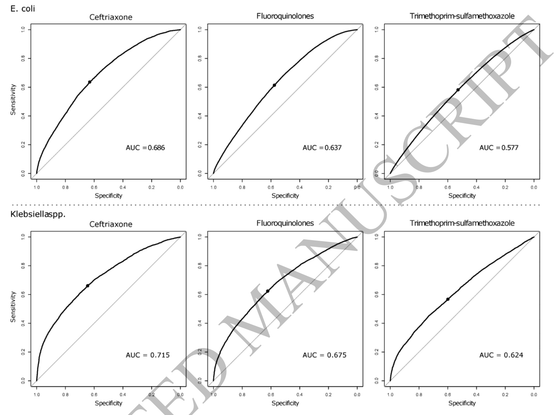
Diagnostic Accuracy of Hospital Antibiograms in Predicting the Risk of Antimicrobial Resistance in Enterobacteriaceae Isolates: A Nationwide Multicenter Evaluation at the Veterans Health Administration
We conducted a comprehensive evaluation of diagnostic accuracies for conventional antibiograms to predict antimicrobial resistance among E. coli and Klebsiella
Taking a “reality-centric” approach to AI - the transformative potential (and challenges) of the technology in areas of healthcare such as personalised medicine and clinical trials.
#pharma #lifesciences #biotech #AI #ArtificialIntelligence #MachineLearning #ClinicalTrials #PersonalisedMedicine #PersonalizedMedicine #PrecisionMedicine