Now published... and on the #BiophysJ cover!
Cell Adhesion Pattern Shows Conserved #Scalings under #Geometrical Control
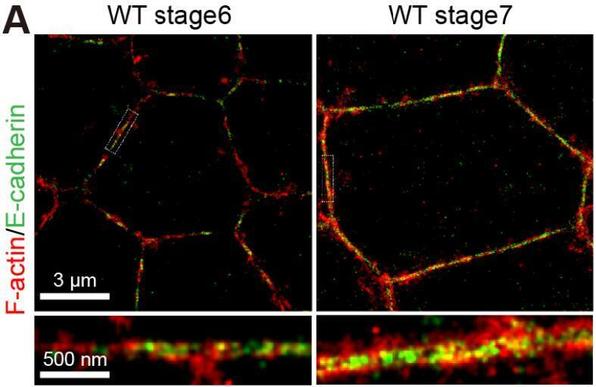
Studying the relationship between the size and the #anatomy and #physiology of #living #organisms has proven useful in deciphering what the key physical constraints are that apply to them. For instance, total adhesive pad area is found to #scale like organism mass over a wide variety of animal species, possibly to resist against body weight.
In this study, we apply this approach at the cell scale to ask how large-scale geometrical constraints affect the organisation of #adhesion patches and the size of #cellSubstrate contact at different stages of the #cellSpreading process. Indeed, #cells, like many animals, adhere to their substrate. For that, they use specific protein complexes, called #focalAdhesions, which form patches often situated at the cell periphery.
cc @JonFouchard
https://www.biophysics.org/blog/cell-adhesion-pattern-shows-conserved-scalings-under-geometrical-control
#cellAdhesion #biophysics #allometry