| https://www.linkedin.com/in/tidona/ | |
| BioMed X | https://bio.mx |
Christian Tidona
- 24 Followers
- 41 Following
- 35 Posts
What if we could study #cancer progression and #metastasis in vivo, in real time, and at single-cell resolution?
David Entenberg, Maja Oktay, and John Condeelis at Albert Einstein College of Medicine summarize the current state of the art of #intravital #imaging, present some of the challenges that remain in the field, how to address them, and future perspectives.
Article in Nature Reviews Cancer: https://www.nature.com/articles/s41568-022-00527-5
Intravital imaging to study cancer progression and metastasis - Nature Reviews Cancer
This Review covers recent advances in intravital imaging of mammalian models of cancer and describes how intravital imaging can help to understand the role of the tumour microenvironment in cancer progression and metastasis, and to develop novel treatments and therapies.
Multispecies biofilm architecture determines bacterial exposure to phages
The spatial dynamics of host-bacteriophage interaction in bacterial biofilms are mostly unexplored at high resolution. This study shows that multispecies bacterial biofilm architecture qualitatively alters the community dynamics of matrix production, interspecific competition, and bacteriophage propagation.
Did you know that during #embryo development, migrating #monocytes deposit tracks of pro-angiogenic #migrasomes guiding the formation of blood vessels?
In this interesting manuscript published in Nature Cell Biology, researchers at Tsinghua University in Beijing studied this phenomenon in the chorioallantoic membrane of chick embryos: https://www.nature.com/articles/s41556-022-01026-3
This could have implications in organotypic #tissueengineering and provide new ways how to inhibit #angiogenesis in solid #tumors.
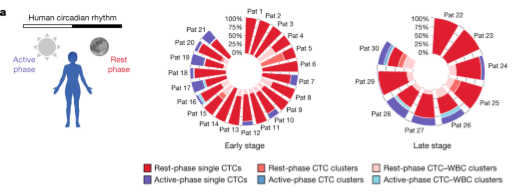
Did you know that circulating #tumor cells (CTCs) which are generated while we #sleep are much more likely to form metastases?
In this very interesting paper, researchers in the lab of Nicola Aceto at ETH Zürich found surprising evidence that the spread of tumors is dependent on the #circadian rythm of the patient.
This means we should consider carefully at which time of the day we diagnose or treat metastasizing cancers.
Article in Nature: https://www.nature.com/articles/s41586-022-04875-y
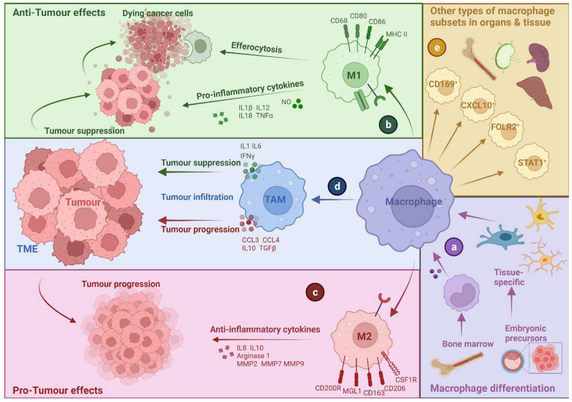
Did you know that dying #cancer cells in solid #tumors can drive tumor-associated #macrophages (TAMs) in both directions? They can become pro-tumorigenic and anti-tumorigenic.
Here is a very nice review article about the interface of TAMs with dying cancer cells in #immunooncology by the lab of Abhishek D. Garg at KU Leuven: https://www.mdpi.com/2073-4409/11/23/3890
#celldeath #efferocytosis #microenvironment #immunosuppression #chemotherapy #radiotherapy
The Interface of Tumour-Associated Macrophages with Dying Cancer Cells in Immuno-Oncology
Tumour-associated macrophages (TAMs) are essential players in the tumour microenvironment (TME) and modulate various pro-tumorigenic functions such as immunosuppression, angiogenesis, cancer cell proliferation, invasion and metastasis, along with resistance to anti-cancer therapies. TAMs also mediate important anti-tumour functions and can clear dying cancer cells via efferocytosis. Thus, not surprisingly, TAMs exhibit heterogeneous activities and functional plasticity depending on the type and context of cancer cell death that they are faced with. This ultimately governs both the pro-tumorigenic and anti-tumorigenic activity of TAMs, making the interface between TAMs and dying cancer cells very important for modulating cancer growth and the efficacy of chemo-radiotherapy or immunotherapy. In this review, we discuss the interface of TAMs with cancer cell death from the perspectives of cell death pathways, TME-driven variations, TAM heterogeneity and cell-death-inducing anti-cancer therapies. We believe that a better understanding of how dying cancer cells influence TAMs can lead to improved combinatorial anti-cancer therapies, especially in combination with TAM-targeting immunotherapies.
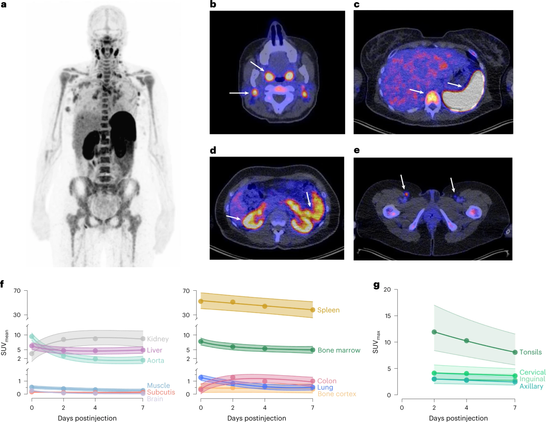
What if we could trace #cytotoxic #Tcells throughout the human body before and during #cancer #immunotherapy?
Researchers at the University of Groningen have used a CD8-specific positron emission tomography (#PET) tracer in a phase 1/2 clinical trial. Patients with solid tumors were imaged before and ~30 days after starting treatment with #antibodies against PD-L1, PD-1, and/or CTLA-4.
Article in Nature Medicine: https://www.nature.com/articles/s41591-022-02084-8
Whole-body CD8+ T cell visualization before and during cancer immunotherapy: a phase 1/2 trial - Nature Medicine
A CD8-specific, one-armed antibody positron emission tomography tracer enables the visualization of the immune response in patients with solid tumors before and after starting immunotherapy.
Did you ever wonder how tissues which have a very high cell turnover rate like in our #intestine control #proliferation to avoid the development of #cancer?
In this very elegant study, Guizela Huelsz Prince and colleagues at AMOLF in #Amsterdam showed in intestinal #organoids that two daughter cells from the same mother cell always follow the same trajectory. Either they both divide, or neither divides. As a result, cell growth is constant.
Article in #eLife: https://elifesciences.org/articles/80682#x080a132e
Mother cells control daughter cell proliferation in intestinal organoids to minimize proliferation fluctuations
Tracking of all cells in intestinal organoids crypts to fully reconstruct cell lineages reveals that proliferative behavior is highly symmetric between sisters cells, while simulations show that this observed symmetry minimizes random fluctuations in cell proliferation, thereby ensuring homeostasis.