That Magic Gene…
While writing yesterday’s post about the evolution of languages, and the importance of genetic information in reconstructing the story thereof, I was reminded of a post I wrote a while ago about the peculiarities of my own genome, a listing of which I have on a CD-ROM at home. There’s not as much data involved as you might think: it’s effectively only about 800 MB.
Anyway, to cut a long story short, it turns out that I have the CCR5-Δ32 genetic mutation. Not only that, I have it twice over, in that I have two copies (homozygotes). I must therefore have inherited it from both parents. This mutation confers virtually complete immunity from HIV-1 infection.
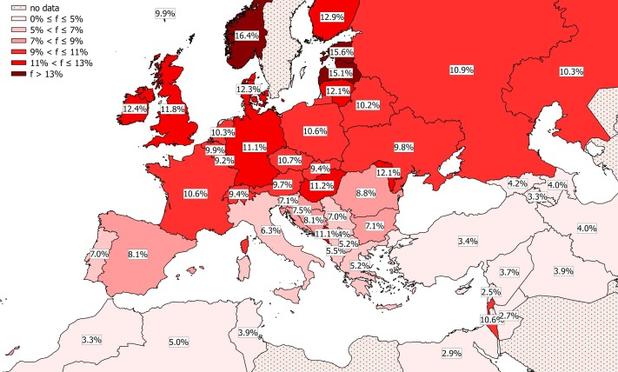
The above graphic shows that more than 10% of the population in Northern Europe has this mutation in just one allele (i.e. they are heterozygotic). To get an estimate of how many have the form on two alleles (i.e. homozygotic) form you can just square that number, so around 1% or more.
It is thought that the CCR5-Δ32 mutation occurred in a single individual in Scandinavia around 1,000 years ago. When I wrote that post I tacitly assumed that it had propagated passively, i.e. without any particular selection, to the modern era. That it reached 10% of the population starting from just one individual surprised me, but I let it pass.
More recently, I came across a paper about how advances in genetics have impacted epidemiological studies. In the abstract it shows that my assumption was probably incorrect.
Algorithms of molecular evolutionary theory suggested that the CCR5-Δ32 mutation occurred but once in the last millennium and rose by strong selective pressure relatively recently to a ~10% allele frequency in Europeans.
It goes on to say this:
Several lines of evidence support the hypothesis that CCR5-Δ32 was selected due to its protective influence to resist Yersinia pestis, the agent of the Black Death/bubonic plague of the 14th century.
I didn’t mention in yesterday’s post that evidence of the plague bacillus is found in a significant number of prehistoric human remains and this almost certainly played a role in the ebb and flow of populations. In the context of CCR5-∆32, however it seems that it may have been advantageous to carry it long before the arrival of HIV/AIDS. That might account for it reaching the relatively high level that it did.
Among the downsides, however, as the article explains, are an increased risk for encephalomyelitis and death when infected with the West Nile virus. Hopefully further cohort studies of people with this mutation will help elucidate its effect on other diseases.
By the way, in contrast to most people I know, I have still never had Covid-19…
 🐦🔥nemo™🐦⬛ 🇺🇦🍉
🐦🔥nemo™🐦⬛ 🇺🇦🍉