What is one phrase in your field of study that appears in nearly every paper introduction? For cellulose biosynthesis, I would have to say it's this:
"Cellulose is the most abundant biopolymer on Earth."
#RNA-protein complexes have amazingly complex architectures. New proteomic and RNA co-purification results from my colleagues indicate the propensity of the eIF2A factor to bind both to mRNA for genes involved in yeast cell wall metabolism and also to proteins involved in RNA degradation.
eIF2A represses cell wall biogenesis gene expression in Saccharomyces cerevisiae - PubMed
Translation initiation is a complex and highly regulated process that represents an important mechanism, controlling gene expression. eIF2A was proposed as an alternative initiation factor, however, its role and biological targets remain to be discovered. To further gain insight into the function of …
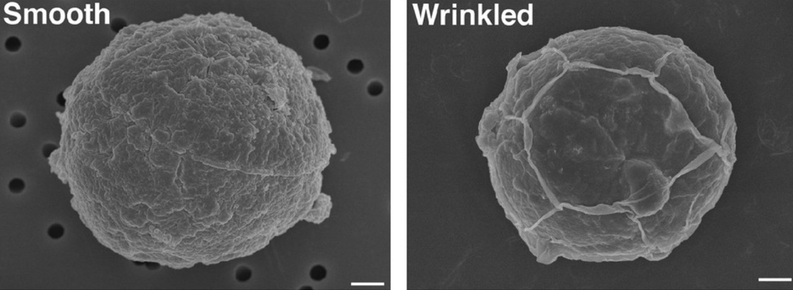
Using a drug without understanding it's mechanism is risky business. Although obvious, some are so useful that questions are swept away in the waves of enthusiastic adoption.
Straight from the keyboard next door, where Mike took the time to ask those questions nonetheless, comes this report that the #CellWall field's favourite cellulose inhibitor, isoxaben, behaves in ways very difficult to reconcile with the assumption that it specifically inhibits CESAs: https://doi.org/10.1093/plphys/kiad538
#Algae living inside #corals provide sugars for their host by digesting their own cell walls https://elifesciences.org/articles/90916 @eLife #protists #microbes #symbiosis
Environmental pH signals the release of monosaccharides from #CellWall in #coral #symbiotic alga https://elifesciences.org/articles/80628
"combination of both digestion- and transporter-driven secretion are likely involved in sugar output... The symbiotic system may therefore be tunable according to the dynamic conditions of the coral reef."
New Review article from the lab of Tuo Wang on #NMR for the study of polysaccharide assemblies in #plant and fungal cell walls:
https://www.sciencedirect.com/science/article/pii/S0969212623002733
🍅Perfect day to talk 🍅#tomatoes with Shi et al. who🍅review #transcriptional regulation of 🍅processes related to 🍅fleshy #fruit texture🔓⬇️🍅https://jipb.net/EN/10.1111/jipb.13316
@wileyplantsci
🍅 #PlantSci #development 🍅#CellWall #TF #PlantScience #MADSBoxProtein
Why does *in vitro* lignin depolymerization by fungal laccases rarely work out? A presentation by Elena Gugole at #ICP2023 in Nantes offered a very satisfying answer:
When fungi degrade lignin, they do so in concert with cellulose degradation, where the phenolic radicals from lignin breakdown are used as electron acceptors by glucose dehydrogenases. *In vitro, *outside the context of cellulose degradation, those radicals re-polymerise instead. #plantscience #cellwall
Crystal structure and functional characterization of an oligosaccharide dehydrogenase from Pycnoporus cinnabarinus provides insights into fungal breakdown of lignocellulose - Biotechnology for Biofuels and Bioproducts
Background Fungal glucose dehydrogenases (GDHs) are FAD-dependent enzymes belonging to the glucose-methanol-choline oxidoreductase superfamily. These enzymes are classified in the “Auxiliary Activity” family 3 (AA3) of the Carbohydrate-Active enZymes database, and more specifically in subfamily AA3_2, that also includes the closely related flavoenzymes aryl-alcohol oxidase and glucose 1-oxidase. Based on sequence similarity to known fungal GDHs, an AA3_2 enzyme active on glucose was identified in the genome of Pycnoporus cinnabarinus, a model Basidiomycete able to completely degrade lignin. Results In our work, substrate screening and functional characterization showed an unexpected preferential activity of this enzyme toward oligosaccharides containing a β(1→3) glycosidic bond, with the highest efficiency observed for the disaccharide laminaribiose. Despite its sequence similarity to GDHs, we defined a novel enzymatic activity, namely oligosaccharide dehydrogenase (ODH), for this enzyme. The crystallographic structures of ODH in the sugar-free form and in complex with glucose and laminaribiose unveiled a peculiar saccharide recognition mechanism which is not shared with previously characterized AA3 oxidoreductases and accounts for ODH preferential activity toward oligosaccharides. The sugar molecules in the active site of ODH are mainly stabilized through CH-π interactions with aromatic residues rather than through hydrogen bonds with highly conserved residues, as observed instead for the fungal glucose dehydrogenases and oxidases characterized to date. Finally, three sugar-binding sites were identified on ODH external surface, which were not previously observed and might be of importance in the physiological scenario. Conclusions Structure–function analysis of ODH is consistent with its role as an auxiliary enzyme in lignocellulose degradation and unveils yet another enzymatic function within the AA3 family of the Carbohydrate-Active enZymes database. Our findings allow deciphering the molecular determinants of substrate binding and provide insight into the physiological role of ODH, opening new perspectives to exploit biodiversity for lignocellulose transformation into fuels and chemicals.
#introduction. I am a plant developmental geneticist. The lab addresses a fundamental biological question: how does morphogenesis translate genotype into form? To this end we study signaling at the plasma membrane - cell wall interface and we investigate how mechanical forces influence tissue shape using 3D digital ovules.
Main interests:
#Arabidopsis #CellBiology #cellbio #CellWall #devbio #devbiol #DevelopmentalBiology #EvoDevo #imaging #lignin #MechanoBiology #microscopy #PlantBiology