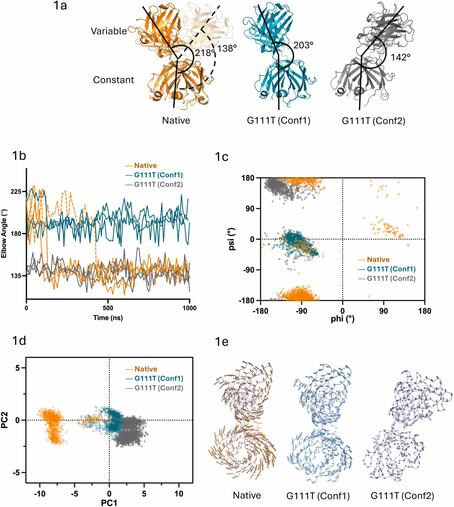
🧬 One amino acid to stabilize them all — myth or molecular reality?
🔗 Single-residue engineering of lambda (λ) antibody light chains reduces conformational flexibility and enhances thermal stability. Computational and Structural Biotechnology Journal, DOI: https://doi.org/10.1016/j.csbj.2025.10.045
📚 CSBJ: https://www.csbj.org/
#MonoclonalAntibodies #mAbs #Immunology #ComputationalBiology #MolecularDynamics #Bioinformatics #ProteinStability #StructuralBiology
RE: https://bsky.app/profile/did:plc:oxh2lmowuusxjqn4f4usyjk5/post/3lkkzp4ocn22q
#drawloween #mabs #dracula #witch
#Escape of #SARS-CoV-2 #variants #KP11, #LB1 and #KP33 from approved #mAbs http://biorxiv.org/cgi/content/short/2024.08.20.608835v1?rss=1
#Sipavibart neutralized JN.1.1 but lost antiviral efficacy against KP1.1, LB.1 and KP3.3. Our results highlight the need for a close clinical monitoring of Pemivibart and raise concerns about the clinical efficacy of Sipavibart.
A Randomized, Placebo-Controlled Trial to Evaluate the Safety and Efficacy of VIR-2482 {#mAbs} in Healthy #Adults for #Prevention of #Influenza A Illness (PENINSULA), Clin Infect Dis.: https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciae368/7717845?login=false
VIR-2482 1200 mg IM was well tolerated but did not significantly prevent protocol-defined ILI. Secondary endpoint analyses suggest this dose may have reduced influenza A illness.
A Randomized, Placebo-Controlled Trial to Evaluate the Safety and Efficacy of VIR-2482 in Healthy Adults for Prevention of Influenza A Illness (PENINSULA)
AbstractBackground. Influenza A results in significant morbidity and mortality. VIR-2482, an engineered human monoclonal antibody with extended half-life,
Antigenicity and receptor affinity of SARS-CoV-2 BA.2.86 spike - Nature
A SARS-CoV-2 Omicron subvariant, BA.2.86, has emerged and spread to numerous countries worldwide, raising alarm because its spike protein contains 34 additional mutations compared to its BA.2 predecessor1. We examined its antigenicity using human sera and monoclonal antibodies (mAbs). Reassuringly, BA.2.86 was not more resistant to human sera than the currently dominant XBB.1.5 and EG.5.1, indicating that the new subvariant would not have a growth advantage in this regard. Importantly, sera from patients who had XBB breakthrough infection exhibited robust neutralizing activity against all viruses tested, suggesting that upcoming XBB.1.5 monovalent vaccines could confer added protection. While BA.2.86 showed greater resistance to mAbs to subdomain 1 (SD1) and receptor-binding domain (RBD) class 2 and 3 epitopes, it was more sensitive to mAbs to class 1 and 4/1 epitopes in the “inner face” of RBD that is exposed only when this domain is in the “up” position. We also identified six new spike mutations that mediate antibody resistance, including E554K that threatens SD1 mAbs in clinical development. The BA.2.86 spike also had a remarkably high receptor affinity. The ultimate trajectory of this new SARS-CoV-2 variant will soon be revealed by continuing surveillance, but its worldwide spread is worrisome.