Interesting ChemRxiv preprint by the group of Katrin Rittinger. They use enantiomeric pairs of covalent inhibitors to profile ligandable cysteines in the proteome. Interesting use of machine learning to predict new compounds that can add most to the ligandable space.
https://doi.org/10.26434/chemrxiv.15002030/v1
#ChemicalProbes #ChemicalProteomics #ProteoProbes #Chemistry #ChemBio #Ligandability #Cysteine #CovalentInhibitor
Very excited that Jon Stokes will visit us on May 28th for a
@[email protected] Lecture. Very much looking forward to learning more about the potential of AI to speed up antibiotic discovery.
#Antibiotics #DrugDiscovery #AI #ChemBio #ChemSkyVery excited that Jon Stokes will visit us on May 28th for a @LED3hub Lecture. Very much looking forward to learning more about the potential of AI to speed up antibiotic discovery.
#Antibiotics #DrugDiscovery #AI #ChemBio #Chemistry
We are looking forward to the LED3 Lecture by Jon Stokes on May 28th. He will talk about "An honest discussion about AI for antibiotic discovery "
If you are interested, make sure to come by!
https://www.universiteitleiden.nl/en/science/led3/led3-lectures
#ChemBio #Chemistry #DrugDiscovery #Antibiotics #AI
Another thought-provoking
#MedChem #SciComm story by Stan Van Boeckel.
On the example of Sitagliptin, he explains the power of minimizing molecules and looking for the highest ligand efficiency in
#DrugDiscovery.
www.linkedin.com/posts/stan-v...
#ChemSky #ChemBio #SmallMolecules #Science #MedicinesPart 29 — Sometimes “smaller i...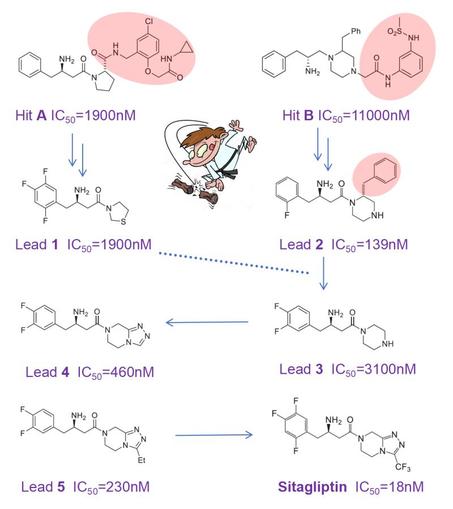
Part 29 — Sometimes “smaller is elegant” in drug discovery.
In early discovery, medicinal chemists often chase potency by making molecules larger and more lipophilic. Yet potency should always be… | Stan Van Boeckel
Part 29 — Sometimes “smaller is elegant” in drug discovery.
In early discovery, medicinal chemists often chase potency by making molecules larger and more lipophilic. Yet potency should always be interpreted in the context of molecular weight and lipophilicity. Ligand‑efficiency metrics normalize potency to size and logP, helping chemists distinguish truly efficient hits and leads from bulky or greasy molecules that only appear potent but usually bring ADMET liabilities. The real challenge is to think broadly, so consider to make your hit or lead smaller during optimization, or 'just do it'.
The discovery of sitagliptin illustrates this beautifully: a blockbuster drug that is smaller than the original screening hits. Sitagliptin (approved 2006) is the first DPP‑4 inhibitor that slows the rapid degradation of GLP‑1 and GIP, thereby lowering plasma glucose. In Merck’s HTS campaign, compounds A and B emerged as low‑potency hits. Here comes the twist: instead of growing these hits, the team chopped off large moieties and explored whether the resulting fragments could be optimized with small substituents such as fluorines. Surprisingly, this yielded compact leads 1 and 2 with improved potency, while selectivity across DPP family members and oral bioavailability in rat were continuously monitored.
Lead 2 was then chopped again-taking inspiration from lead 1, which lacked its extra phenyl ring-to give weak lead 3. Accepting a 20‑fold drop in IC₅₀ seems counterintuitive, but it opened the door to expand the piperazine and create a new heterocycle. This delivered more potent lead 4, followed by lead 5. Further fluorination of 5 improved oral exposure and ultimately delivered the blockbuster sitagliptin.
This small, well‑balanced molecule (MW 407; IC₅₀ 18 nM; oral bioavailability 87%; t½ 12 h; rapid absorption; logP 1.5; high selectivity; medium solubility; 5 rotatable bonds; HA/HD 4/1; non‑toxic) is a superb example of optimizing property space without molecular obesity. A remarkable achievement by Malcolm MacCoss, Ph.D., FRSC and his team at Merck.

Part 29 — Sometimes “smaller is elegant” in drug discovery.
In early discovery, medicinal chemists often chase potency by making molecules larger and more lipophilic. Yet potency should always be… | Stan Van Boeckel
Part 29 — Sometimes “smaller is elegant” in drug discovery.
In early discovery, medicinal chemists often chase potency by making molecules larger and more lipophilic. Yet potency should always be interpreted in the context of molecular weight and lipophilicity. Ligand‑efficiency metrics normalize potency to size and logP, helping chemists distinguish truly efficient hits and leads from bulky or greasy molecules that only appear potent but usually bring ADMET liabilities. The real challenge is to think broadly, so consider to make your hit or lead smaller during optimization, or 'just do it'.
The discovery of sitagliptin illustrates this beautifully: a blockbuster drug that is smaller than the original screening hits. Sitagliptin (approved 2006) is the first DPP‑4 inhibitor that slows the rapid degradation of GLP‑1 and GIP, thereby lowering plasma glucose. In Merck’s HTS campaign, compounds A and B emerged as low‑potency hits. Here comes the twist: instead of growing these hits, the team chopped off large moieties and explored whether the resulting fragments could be optimized with small substituents such as fluorines. Surprisingly, this yielded compact leads 1 and 2 with improved potency, while selectivity across DPP family members and oral bioavailability in rat were continuously monitored.
Lead 2 was then chopped again-taking inspiration from lead 1, which lacked its extra phenyl ring-to give weak lead 3. Accepting a 20‑fold drop in IC₅₀ seems counterintuitive, but it opened the door to expand the piperazine and create a new heterocycle. This delivered more potent lead 4, followed by lead 5. Further fluorination of 5 improved oral exposure and ultimately delivered the blockbuster sitagliptin.
This small, well‑balanced molecule (MW 407; IC₅₀ 18 nM; oral bioavailability 87%; t½ 12 h; rapid absorption; logP 1.5; high selectivity; medium solubility; 5 rotatable bonds; HA/HD 4/1; non‑toxic) is a superb example of optimizing property space without molecular obesity. A remarkable achievement by Malcolm MacCoss, Ph.D., FRSC and his team at Merck.
Join us as member of the Division of Biochemistry of the @gdch!
- A vibrant network of biochemists
- Connection with international experts
- Reduced fees for division conferences
- Support of junior scientists via travel grants and prizes
https://en.gdch.de/network-structures/gdch-structures/division-member.html?tx_powermail_pi1%5Bfield%5D%5Bfachgruppe%5D=Biochemie
#Biochemistry #ChemBio #Chemistry
We are again looking for 3 Flash Talks for the 6th Virtual @ChemBioTalks. If you are a PhD candidate or postdoc in the field of #ChemBio, I strongly encourage you to apply via our abstract challenge: https://chemistry-europe.onlinelibrary.wiley.com/hub/journal/14397633/chembio-flash
#ChemBio #Chemistry #Competition






