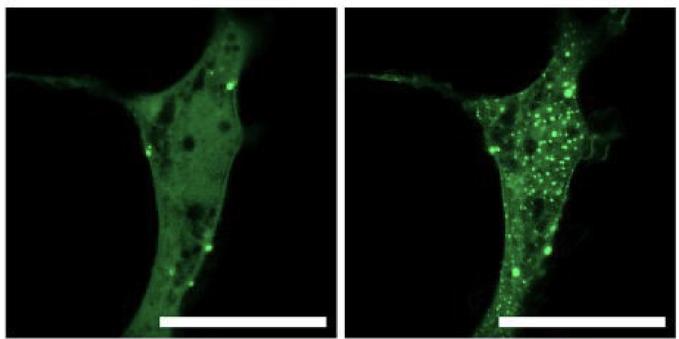
Please share …. 🙏
📣 Very soon, we’ll have an open PhD position starting in fall 2025 — this time not on cilia, but on YAP & TAZ in podocytes and their regulation via LLPS. Interested candidates are welcome to contact us directly.
Supervision by Inês Cabrita and me, embedded within PodoSigN!
Within this project, close collab. with Beate Vollenbröker‘s team.
#Podocyte #Trr422 #Fsgs #Nephrolab #PhdPosition #Biology #Genetics #LLPS #HippoSignaling #phd
Two new articles from our lab:
Calcium-Dependent S100A8 Amyloid Fibril Formation via S100A1-Mediated Transient Interaction https://pubs.acs.org/doi/10.1021/acschemneuro.5c00086
Heterotypic Droplet Formation by Pro-Inflammatory S100A9 and Neurodegenerative Disease-Related α-Synuclein https://pubs.acs.org/doi/10.1021/acs.biomac.5c00130
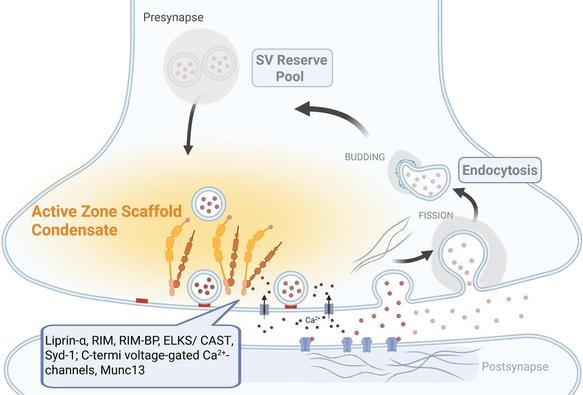
#Cellular structure without a #membrane: Researcher discusses how #synapses use #liquids to create functional separations.
#LLPS #membrane-less_organelles #neurotransmission
https://phys.org/news/2025-03-cellular-membrane-discusses-synapses-liquids.html
Cellular structure without a membrane: Researcher discusses how synapses use liquids to create functional separations
The laboratory of Ege Kavalali, professor and chair of the Department of Pharmacology, published a paper in Nature Communications that determined that liquid-liquid phase separation plays a key role within the nanostructure of synapses, and that its disruption affects evoked but not spontaneous neurotransmission.
Check out our new preprint titled "Diverse effects of fluorescent labels on alpha-synuclein condensate formation during liquid-liquid phase separation"
Hello Mastodon,
Our first post is about Liquid-liquid phase separation (LLPS) phenomena, during which proteins form liquid droplets, followed by amyloid aggregation. LLPS is observed in neurodegenerative disorders, cancers and certain cases is related to protein functions in the cell nucleus. Watch below as alpha-synuclein, a protein linked to Parkinson's disease, undergoes LLPS.
FAM81A is a postsynaptic protein that regulates the condensation of postsynaptic proteins via liquid–liquid phase separation
Proteomic analyses of postsynaptic proteins have mostly overlooked those with poorly characterized functions, likely missing crucial contributors to synaptic biology. Through a meta-analysis the authors identify 97 postsynaptic proteins with poorly characterized function, and elucidate the role FAM81A, revealing that it regulates the condensation of postsynaptic proteins via liquid-liquid phase separation to affect the size and function of synapses.
RNA-mediated ribonucleoprotein assembly controls TDP-43 nuclear retention
TDP-43 nuclear localization is crucial for its function during RNA processing and pathogenesis of neurodegenerative disorders. This manuscript demonstrates that RNA binding and macromolecular assembly, through multivalent interactions and liquid-liquid phase separation, play a central role in TDP-43 nuclear retention.