Overcoming vascular niche–mediated TKI resistance in acute myeloid leukemia through miR-126 inhibition
#Cancer #JanusEffect #AML #ABM
https://www.nature.com/articles/s41540-026-00675-6?utm_source=rct_congratemailt&utm_medium=email&utm_campaign=oa_20260320&utm_content=10.1038/s41540-026-00675-6
Overcoming vascular niche–mediated TKI resistance in acute myeloid leukemia through miR-126 inhibition - npj Systems Biology and Applications
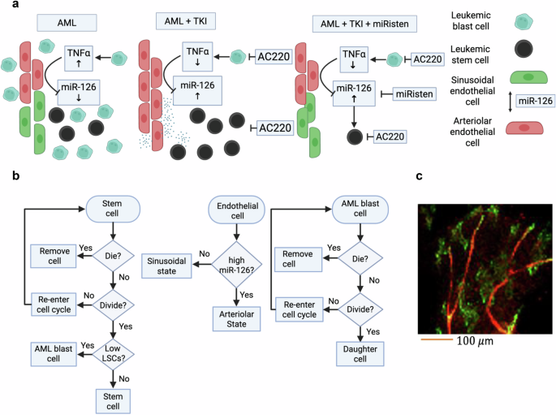
Acute myeloid leukemia (AML) is a hematologic malignancy originating in the bone marrow and often progressing to extramedullary sites. Despite advances in molecularly targeted therapies and hematopoietic stem cell transplantation, clinical outcomes remain poor. Tyrosine kinase inhibitors (TKIs) provide benefit to a subset of AML patients harboring FLT3-ITD mutations; however, relapse and resistance remain common. These therapeutic failures are driven by both intrinsic properties of leukemic stem cells (LSCs)—a quiescent, self-renewing population—and extrinsic cues from the tumor microenvironment. We previously demonstrated that arteriolar endothelial cells (ECs) produce miR-126, which is transferred to LSCs, promoting quiescence, treatment resistance, and niche retention. During disease progression, TNF-α secreted by expanding blasts suppresses EC miR-126 production. Following TKI administration, blast reduction lowers TNF-ɑ levels, restoring EC miR-126 production, and this miR-126 expression enables LSCs to re-enter quiescence—thereby escaping therapy and facilitating relapse. To explore this dynamic, we developed an agent-based computational model of the AML bone marrow microenvironment, parameterized with in vitro and in vivo data. The model captures vascular niche remodeling and feedback between leukemic populations and endothelial signaling. Simulations reveal that LSC protection mediated by miR-126 can be disrupted by combining TKIs with miRisten, a miR-126 inhibitor. When administered on a defined schedule, this combination dismantles the protective niche and enhances LSC eradication. These findings underscore the therapeutic potential of targeting microenvironmental feedback to overcome resistance and prevent AML relapse.