Cephalic furrow thread
I published a cephalic furrow thread about our paper on Twitter and Mastodon. We recently updated the manuscript on bioRxiv. In addition to the tissue mechanics, this new version includes gene expression data comparing Drosophila with Clogmia, a fly that has no cephalic furrow… It gives us some hints about the patterning changes associated with the evolution of this novel invagination.
Screenshot of the cephalic furrow paper on bioRxiv.
https://doi.org/10.1101/2023.03.30.534554 1. Opening
Our paper on cephalic furrow mechanics got updated with an exciting evo-devo twist :)
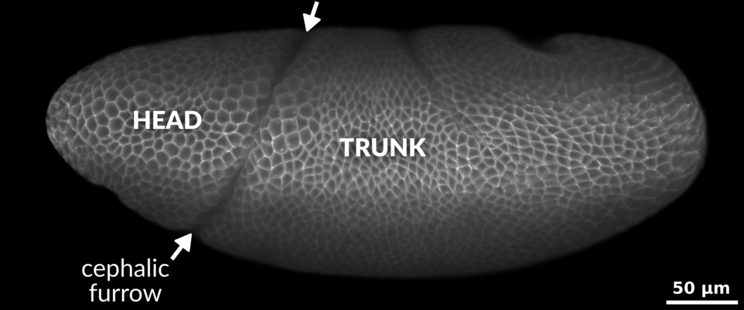
Lateral view of a
Drosophila embryo during gastrulation. Between the head (left) and the trunk (right) lies a prominent epithelial invagination extending from dorsal to ventral, known as the cephalic furrow (marked by arrows).
From the labs of Pavel Tomancak and Carl Modes:
What can a fold tell us about the interplay between genetic patterning, tissue mechanics, and the evolution of morphogenesis?
https://www.biorxiv.org/content/10.1101/2023.03.30.534554v2
2. What’s the role?
The cephalic furrow is a puzzling fold.
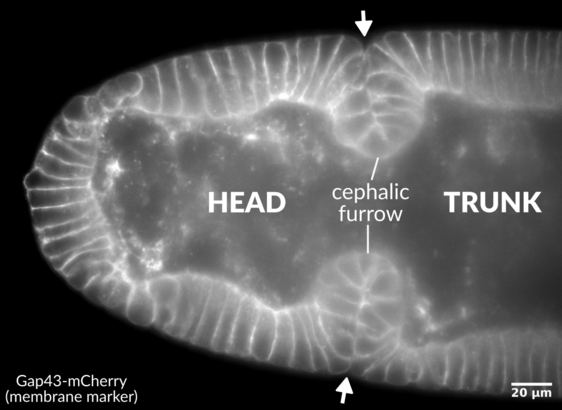
Profile view of the anterior end of a
Drosophila embryo, showing the bilateral cephalic furrow invaginations between the head and trunk (arrows).
While its formation is under strict genetic control, the invagination is transient and doesn’t give rise to specific tissues. It simply unfolds, leaving no trace.
It has no obvious function. Or does it?
3. Instability in mutants
To investigate, we analyzed the tissue dynamics in cephalic furrow mutants.
[
DOWNLOAD VIDEO] Time-lapse movie of two
Drosophila embryos in lateral view during gastrulation. The top embryo is a control and the bottom embryo is a
buttonhead mutant. In
btd mutants, the cephalic furrow fails to form. Instead, ectopic folds appear around the head-trunk interface as a result of mechanical instability on the epithelium.
We found that without the invagination, the head-trunk epithelium becomes unstable and buckles, forming what we call ectopic folds (see bottom embryo).
What’s causing this instability?
4. Sources of stress
We identified two potential sources of mechanical stress…
Lateral view of a cephalic furrow mutant, showing schematic drawings of two sources of mechanical stress during gastrulation: mitotic domains and germ band extension. Mitotic domains undergo apical area expansion during cell division, and ectopic folds form in between and adjacent to them.
Mitotic domains (groups of dividing cells in the head) and germ band extension (tissue movement towards the head).
…and analyzed their contribution using biophysical modeling and experimental perturbations.
5. Biophysical model
Profile view of a
Drosophila embryo showing the traits we used to build a biophysical model of the epithelium with particles connected by springs.
The model developed by Abhijeet Krishna and Alicja Szałapak from the Carl Modes lab allowed us to simulate epithelial dynamics in different conditions, and make predictions for what’s happening in vivo.
Example parameter sweep for our cephalic furrow model. It shows a plot containing representative samples of each simulated condition. A sample shows the epithelium in lateral view and the presence or not of ectopic folds. Mitotic domains are highlighted in pink.
For example, would ectopic folds appear if there was no germ band extension?
6. Germ band cauterization
To test this, Marina Cuenca mechanically blocked the germ band extension by laser cauterization in cephalic furrow mutants. No ectopic folds appear.
[
DOWNLOAD VIDEO] Cauterization experiment to block the extension of the germ band in a cephalic furrow mutant embryo. The top embryo is an
eve mutant, showing the formation of ectopic folds on the head-trunk interface. The bottom embryo is an
eve mutant with a cauterized germ band. No ectopic folds appear.
This indicates that mitotic domains alone cannot induce buckling, only when combined with the germ band push.
7. One double mutant
Similarly, inhibiting mitotic domains in cephalic furrow mutants (double mutant btd–stg) also abolishes ectopic folds.
[
DOWNLOAD VIDEO] Profile views of a regular
btd mutant showing the formation of ectopic folds (top) and a double
btd–
stg mutant showing no ectopic folds (bottom).
This indicates that germ band extension alone cannot induce buckling, only when combined with mitotic domains.
8. Head–trunk under stress
These data suggests that, without the cephalic furrow, the simultaneous formation of mitotic domains and germ band extension generates mechanical instability.
Drosophila embryo in lateral view. Mitotic domains and germ band are illustrated as sources of tissue compression and epithelial instability.
Could the cephalic furrow be counteracting these increased compressive stresses during gastrulation?
9. Early fold prevents buckling
Our simulations point in this direction, showing that an early head invagination can effectively absorb these stresses and prevent epithelial instabilities at the head-trunk boundary.
Example of simulation output from our cephalic furrow model, showing that an early head invagination effectively prevents ectopic folding in the epithelium. Without the cephalic furrow (left) ectopic folds form at variable positions along the head-trunk interface. With the cephalic furrow (right) the single invariant invagination inhibits nearby folding.
10. A mechanical role
Putting all the data together, we propose the cephalic furrow has a function, it plays a mechanical role during Drosophila gastrulation.
This raised an intriguing evolutionary question for us: Could the cephalic furrow have evolved in response to these mechanical stresses?
11. Evolutionary novelty
We were lucky that Steffen Lemke and Yu-Chiun Wang had been working on the cephalic furrow from an evolutionary perspective because they reveal a crucial insight:
The cephalic furrow is an evolutionary novelty limited to a derived group of dipteran flies.
https://www.biorxiv.org/content/10.1101/2023.10.09.561568v1
12. Instability impacts development
They also provide evidence that other flies experience increased head-trunk stresses, and that this instability may impact developmental robustness, supporting the idea that the cephalic furrow evolved in response to mechanical conflict. Their thread:
https://twitter.com/VerenaJKaul/status/1717472879850528915
13. Patterning evolution
Now, we wondered.
The cephalic furrow is a patterned fold. The initiator cells driving the invagination are specified genetically by the narrow overlap between btd and eve domains.
Gene expression pattern overlap between
buttonhead (
btd) and
even skipped (
eve) at the head-trunk boundary specifies the row of initiator cells for the cephalic furrow. The gene
sloppy paired (
slp) delimits the adjacent cells and edges of the cephalic furrow.
So, which genetic changes are associated with the evolution of this novel invagination?
14. Novel head-trunk domain of btd
We compared gene expression between Drosophila and Clogmia, a fly that has no cephalic furrow.
Gene expression comparison between
Drosophila and
Clogmia, a fly that has no cephalic furrow. Expression of
btd (magenta),
eve (orange),
slp (cyan), and
dapi (gray) on the anterior end. The main difference is that
Clogmia lacks the head-trunk domain of
btd present in
Drosophila.
Clogmia lacks btd expression at the head-trunk, a domain crucial for cephalic furrow formation, suggesting the appearance of this domain was a key event in cephalic furrow evolution.
15. Evolutionary scenario
Altogether, the integrative scenario we are establishing is that mechanical instability acted as a selective pressure for the evolution of the cephalic furrow, and that this occurred through the cooption of a new molecular player at the head-trunk boundary.
Schematic illustration summarizing the position of cephalic furrow traits (genetics and mechanics) in a phylogenetic tree. On the right, an evolutionary scenario for the origin of the cephalic furrow, where mechanical stresses as a selective pressure for the evolution of patterned morphogenetic innovations.
16. Mechanoevolution
To us, the cephalic furrow case illustrates an example of a possibly more widespread mechanism on how mechanical forces can influence the evolution of patterned morphogenetic innovations in early development.
17. Thank yous
I’d like to thank Pavel Tomancak, Marina Cuenca, Abhijeet Krishna, Alicja Szałapak, Carl Modes for their crucial contributions, the MPI-CBG facilities for support, and the Steffen Lemke and Yu-Chiun Wang teams for this unique collective effort on such ephemeral but remarkable invagination ;)
18. Data availability
For more the details: https://www.biorxiv.org/content/10.1101/2023.03.30.534554v2
For high-res figures/videos: https://zenodo.org/record/7781916
For main data/analyses: https://zenodo.org/record/7781947
For model/simulations: https://zenodo.org/record/7784906
—
URL: https://brunovellutini.com/posts/cephalic-furrow-thread/
#cephalicFurrow #clogmiaAlbipunctata #diptera #drosophilaMelanogaster #embryo #evoDevo #mechanobiology #postdoc #preprint #publication #tweet