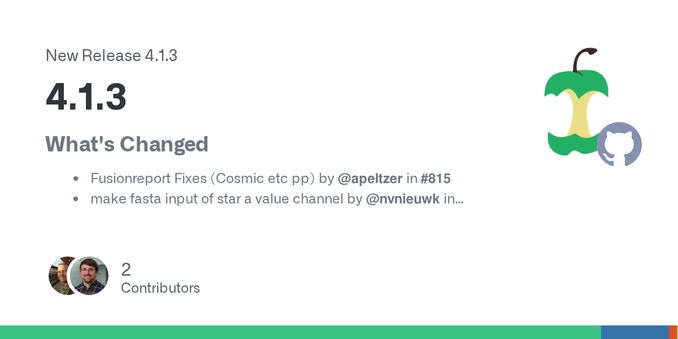
Pipeline release! nf-core/rnavar v1.3.0 - nf-core/rnavar 1.3.0 - Silent Nostromo!
gatk4 RNA variant calling pipeline
Please see the changelog: https://github.com/nf-core/rnavar/releases/tag/1.3.0
#gatk4 #rna #rnaseq #variantcalling #worflow #nfcore #openscience #nextflow #bioinformatics