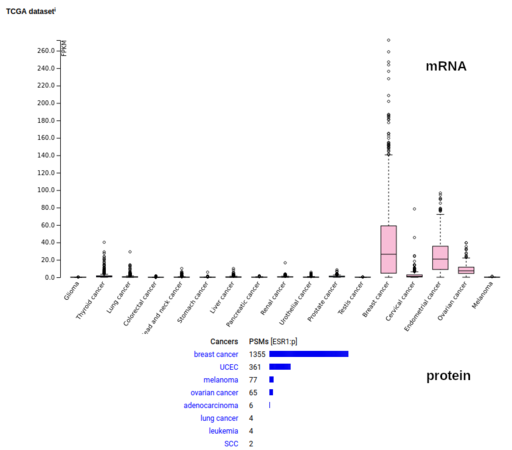
FDA Greenlights New Breast Cancer Therapy, Amidst Data Scrutiny
The FDA approved Veppanu for advanced breast cancer patients with ESR1 mutations who failed prior hormone therapy. Learn what this means for treatment.
#BreastCancer, #Veppanu, #FDA, #CancerTreatment, #ESR1
https://newsletter.tf/fda-approves-veppanu-breast-cancer-esr1-mutation/