Researchers observe how the flexibility of a #protein_hinge is crucial to the transfer of #cell proteins.
https://phys.org/news/2024-01-flexibility-protein-hinge-crucial-cell.html
Researchers observe how the flexibility of a protein hinge is crucial to the transfer of cell proteins
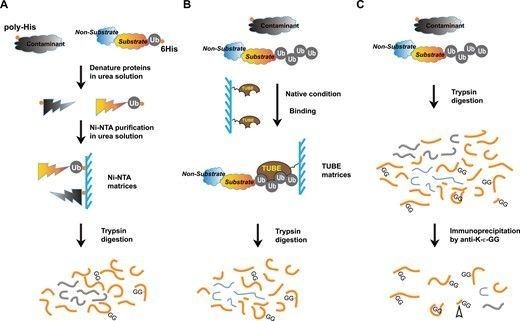
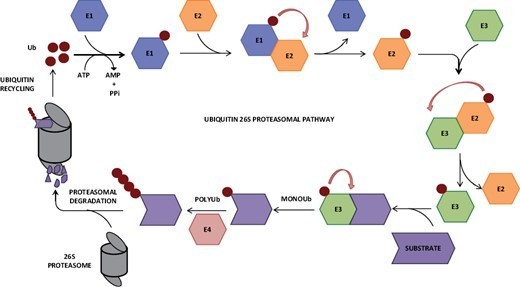
Ubiquitination—the addition of the protein ubiquitin—is a key stage in many cell processes, such as protein degradation, DNA repairs, and signal transduction. Using high-speed atomic force microscopy (HS-AFM) and molecular modeling, researchers led by Hiroki Konno and Holger Flechsig at WPI-NanoLSI, Kanazawa University have identified how the mobility of a ubiquitination-related enzyme hinge allows ubiquitination to take place.