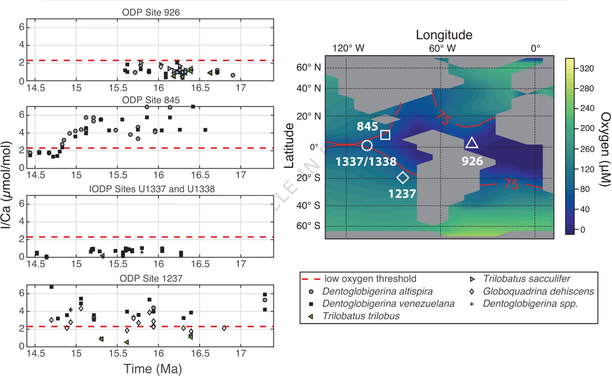
#WeekendReading: Burke et al., on oxygen deficiency in the Atlantic during the Miocene. We're slowly building a nice global picture of the oxygen minimum zones through that period, and their relations to planetary temperatures.
#Cyanobacteria may have been responsible for a mass #extinction event about 2.4 billion years ago.

One of the earliest great extinctions in Earth's history may have been caused not by an asteroid or a volcano but by oxygen itself, when tiny photosynthetic microbes slowly filled the air with a gas that was poison to much of the anaerobic life that ruled the planet long before us
Around 2.4 billion years ago, the air over Earth began to change. Microbes in the oceans, the cyanobacteria, had been running a chemical reaction that split water and released oxygen as waste. For a long time that oxygen was mopped up almost as fast as it was made. Then the sinks filled, and it began […]
https://phys.org/news/2026-05-earth-oxygen-rich-atmosphere-owe.html?utm_source=nwletter&utm_medium=email&utm_campaign=daily-nwletter
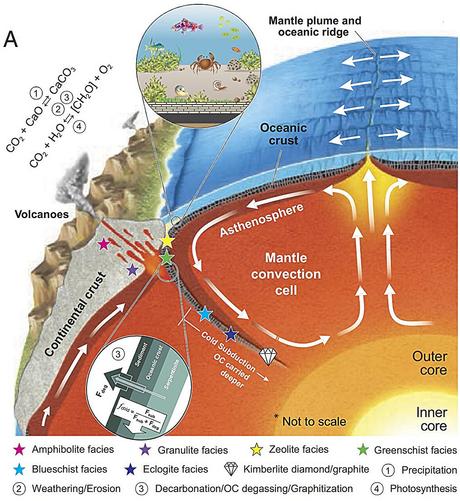
Earth's oxygen-rich atmosphere may owe its existence to cold subduction
Earth was mostly devoid of oxygen for much of its 4.5 billion year lifetime. That is, until certain processes started to allow for the eventual buildup of oxygen up to the levels we have now (around 21% of the atmosphere). While scientists have found evidence of the approximate timescales of rises in oxygen over time and are aware of some of the mechanisms behind it, the main driver behind Earth's long-term oxygenation is still unclear.
https://atlas.whatip.xyz/post.php?slug=does-extracting-oxygen-from-lunar-soil-matter-science
<p>Oxygen from lunar soil as a base building priority A new discussion around space exploration focuses on
#extracting #science #oxygen #matter
Shaunna Burke has summitted Mount Everest with Stage 4 cancer, raising funds to support research into the link between oxygen and cancer progression.
#Health #Cancer #exerciseandcancer #MountEverest
https://globalnews.ca/news/11873619/woman-stage-4-cancer-summit-mount-everest/
Shaunna Burke has summitted Mount Everest with Stage 4 cancer, raising funds to support research into the link between oxygen and cancer progression.
#Health #Cancer #exerciseandcancer #MountEverest
https://globalnews.ca/news/11873619/woman-stage-4-cancer-summit-mount-everest/
Chapter 4 21st century botany gets Classical
Earth, fire, air and water – the four elements of the Ancients – each have important contributions to make in the biology of plants, whether present in the right amount, in excess or insufficiency. The effects of evolution in an environment where water is scarce is shown in the spines of this cactus. The spines are highly modified leaves which significantly reduce uncontrolled loss of water from the plant.
Chapter 4 emphasises the relative simplicity of the fundamental ‘ingredients’ – minerals from the soil, light and heat from the sun, oxygen and carbon dioxide from the atmosphere, and water – that are needed to sustain plant life (even if a few liberties have been taken with modern-day interpretations of the ancient notion of the four ‘elements’). And that is also a testament to the remarkable adaptability of plants, not only to survive, but to thrive, in those environments where there is either an insufficiency or an excess of one or other of those four basic ingredients.
Botany in the 21st century is a sophisticated scientific discipline that has generated a tremendous body of knowledge about how plants work. Whilst we are rightly proud of our present day botanical understanding, we should always be mindful of the roots of that knowledge. In particular, we should respect the early foundations laid down by our ancestors as they attempted to understand plant life and which subsequent generations of scholars have built upon. So, whilst it may be tempting to laugh at some of the quaint notions of times long gone by, with the benefit of our more up-to-date viewpoint, we should try and value them as genuine and sincere attempts to achieve what we are still trying to arrive at today, an understanding of the true nature of plants. In an attempt to show a connection between ancient and modern views of botanical knowledge this essay considers how one ancient notion on the nature of plants, although long since abandoned in its specifics, still has great resonance with a modern understanding of plant biology.
Thousands of years ago – long before it was recognised that living (and non-living) entities were composed of chemical elements (Bruce Alberts et al., 2002; Philip Ball, 2002), philosophers attempted to understand what plants – and all other living things – were made of. One idea widespread in Ancient – so-called Classical – times was the concept that all terrestrial items were composed of four ‘elements’ – earth, fire, air, and water (Benjamin Jowett, 1998; Philip Ball, 2002; John Finney, 2015). Although this notion has long since been discarded in favour of a more scientific understanding of the composition of matter and lifeforms that are composed of that matter, it still has great relevance to our understanding of plant structure today. In fact, arguably, all you need to make a plant is earth, fire, air and water. How so?
Earth
For plants, as at March in 2026 CE, 17 chemical elements are considered essential (Michael A Grusak et al., 2016; Kaleigh Remick & John D Helmann, 2023). Essential means that the elements are: necessary for completion of the entire life cycle – from spore-to-spore (or seed-to-seed for gymnosperms and angiosperms); act within the plant body; and cannot be substituted by another element (Cristie Preston; George Blake et al., 2008).
Needed in relatively large amounts are the so-called macronutrients – carbon, hydrogen, oxygen, phosphorus, potassium, nitrogen, sulphur, calcium, and magnesium; required in lesser amounts are molybdenum, nickel, copper, zinc, manganese, boron, iron, and chlorine – the micronutrients. Of those, carbon (as carbon dioxide) and oxygen (as molecular oxygen) are primarily obtained from the atmosphere (the ‘air’), hydrogen is obtained from water (H2O), primarily within the soil; the other 14 are principally extracted by plants from the soil – which we can interpret as the ‘earth’.
Combined in different ways those 17 elements comprise macromolecules such as proteins, fats, nucleic acids, and polysaccharides – which are employed in the construction of many of the structural components of plant cells and their numerous membranes, and in their turn are arranged as tissues and organs and ultimately the entire plant body. Many of the metals are used as activators of enzymes (Thomas E Hemmerly, 2023; Abdel El Hadrami) helping to ensure that the biochemistry necessary for the plant’s proper functioning operates properly, or are involved in electron transport chains (Thomas E Hemmerly, 2023; Kaider Leila, 2024) to ensure the plant can make sufficient energy, either via photosynthesis in chloroplasts or by respiration in mitochondria (Melissa Ha et al.; Jose Chen Lopez).
The roles played by these essential nutrients are many and varied; all are essential. Some plants need elements additional to those 17 – and which are again primarily derived from the soil – to enable particular processes to take place. For example, CAM [Crassulacean Acid Metabolism] plants need sodium (Ulrich Lüttge, 2015), root-nodulating legumes need cobalt for that mutually beneficial symbiosis to develop (Shaukat-Ahmed & Harold J Evans, 1961), and silicon is crucial to the development of the frustules that surround diatoms (Mark Hildebrand et al., 2018; Carole C Perry, 2022; ) [Ed. – although diatoms are unicellular algae, their photosynthetic capacity (Benoît Schoefs et al., 2017; Claudia Büchel, 2019) accords them the status of ‘honorary plants’ for the purposes of this essay].
Although soil is extremely influential and beneficial to plants in a direct nutritional way, it also performs other important roles in the life of the plant, such as the provision of a rooting medium for the plant – the majority of plants are rooted in the soil rather than having a care-free, rolling-stone existence as for tumbleweed. Soil also acts as a seed bank (PJ Christoffoleti & RSX Caetano, 1998). More globally, it is the combination of latitude (hence temperature and rainfall – ‘fire and water’…) with soil that largely dictates the vegetation type that prevails and hence the ecology of an area (Andrew Scott Medeiros & Taly Dawn Drezner, 2012; Priscyla Maria Silva Rodrigues et al., 2018), thereby contributing to the concept of terrestrial biomes (Irwin N Forseth, 2010).
And, in an evolutionary sense, the Earth sustained the early experiments at land-living when those first plants made the leap from an aquatic existence to a terrestrial one (Mitchell Cruzan, 2019; Jan de Vries & John M Archibald, 2018). Exploiting the intimate relationship that plants have with the soil is the human practice of phytoremediation, “a remediation method that utilizes plants to remove, transfer, or stabilize contaminants in soil and water, leveraging their natural biological processes to improve site characterization and treatment outcomes”.
Fire
The Sun is the ultimate ‘fire’ in the life of plants. But that star produces much more than fire – the heating wavelengths of the electromagnetic spectrum such as infra-red (Kendric C Smith, 2014b). Accordingly, we can interpret fire as being the full range of solar wavelengths, which includes light (visible wavelengths), and several ‘invisible’ wavelengths, both longer and shorter than those in the PAR [Photosynthetically-Active Radiation of wavelengths between 400 and 700 nm (Arman Pazuki et al., 2017; Eric Runkle)] range – e.g., ultra-violet radiation (Linda Chalker-Scott, 2012; Kendric C Smith, 2014a). The sun is thus largely responsible for providing a suitable temperature range within which life-sustaining biochemical reactions can take place in plants (and other organisms).
The light the sun provides is extremely important to plant life; differences in both its ‘quality’ and quantity are amongst the most influential abiotic factors that mediate plant development and behaviour (Rita Teresa Teixeira, 2020). This important role of light is exemplified in the range of plant biological processes with the prefix ‘photo-’: – e.g., photosynthesis (Matthew Johnson, 2017; Tiffany Lui); phototropism (directional growth of plant parts in response to a light stimulus) (Jennifer Holland et al., 2009; Shannon Trueman; Charlotte Gommers, 2020); photomorphogenesis (the different forms that plants take in different light environments(James Shinkle, 2008; Keera A Franklin & James R Shinkle, 2009); and photoperiodism (“how plants regulate their flowering and other developmental processes based on the lengths of day and night, known as photoperiods” (DR Gossett, 2023) (John W Kimball; Tom Buckley).
And fire, in a more literal sense, has dramatically helped to shape the present day vegetation cover of the Earth (WJ Bond et al., 2003, 2005; Juli Pausas & Jon Keeley, 2009; Stephen Pyne, 2010; Andrew C Scott et al., 2014), thereby helping to form habitats and much larger units of vegetation. Maybe as a consequence of this, many plant species are known to require fire-generated smoke or compounds found therein – e.g., karrikins – to assist germination (David C Nelson et al., 2012). Indeed, fire has been posited as a major driver of plant evolution (e.g., Susana Gómez-González et al., 2011; Tianhua He et al., 2011; Juli Pausas et al., 2017; Tianhua He & Byron Lamont, 2018), and of Earth’s ecology (Stephen Pyne, 2010) and biodiversity (Tianhua He et al., 2019) more generally.
Air
The current composition of Earth’s atmosphere (the air) is: diatomic nitrogen (N2), 78.08%; diatomic oxygen (O2), 20.95%; argon (A), 0.93%; and carbon dioxide (CO2), 0.04%. Water (H2O) is also present and ranges from 0 – 4%. Noble gases – such as neon (Ne), helium (He), and krypton (Kr) – and other constituents such as nitrogen oxides (NOx), compounds of sulphur (S), and ozone (O3) are found in very small amounts (Roger A Pielke). The air provides plants with two of their 17 essential nutrients – carbon in the form of carbon dioxide which is fixed into organic compounds for plant growth by photosynthesis, and oxygen that supports aerobic respiration.
Although the air is generally life-sustaining for plants, it can also contain many damaging chemicals, which either form in the atmosphere or are carried long distances by air movements – e.g., O3 (ozone), SO2 (sulphur dioxide) and NOx (various oxides of N) (Simon JM Caporn, 2013). Notoriously, NOx and SO2 combine with water in the atmosphere to produce nitric and sulphuric acid, respectively, which both contribute to damaging ‘acid rain’ (Gene E likens & Thomas J Butler). Its deleterious environmental effects aside, but importantly, such acid rain also adds the essential macronutrients nitrogen and sulphur to water bodies and soil where it falls to earth.
Air – particularly when it moves as wind – is exploited by anemophilous plants as a medium to transfer pollen (Jannice Friedman & Spencer CH Barrett, 2009) from stamen to stigma in sexual reproduction, to carry pheromone-like chemicals produced by certain plants and which entice male insects to attempt copulation with their flowers and thereby facilitate pollination (Heather M Whitney & Beverley J Glover, 2013), or to transport volatile chemical signals to warn unaffected neighbouring plants following herbivore attack (C Kost & M Heil, 2006; Michael Wink, 2016) or human-induced damage (Richard Karban et al., 2000).
And wind is an abiotic factor that can help to sculpt the shape of plants (J Oliver, 1960), thereby providing a dramatic and graphic example of the way in which plants can be ‘formed from’ air. This phenomenon is known as thigmomorphogenesis [“control of plant growth by mechanical stimulation” – Frederik Börnke & Thorsten Rocksch, 2018)] (Frank Telewski, 2016; Janet Braam & E Wassim Chehab, 2017; Danilo D Fernando, 2023). [Ed. – although more usually associated with wind, thigmomorphogenesis can also be seen in aquatic plants as a growth response to interaction between the plants and the water that surrounds them (Jonas Schoelynck et al., 2015)] Wind is an important ecological factor in the lives of many plants (AR Ennos, 1997), whose effects extend from direct interactions such as leaf flutter to tree-uprooting and seed dispersal, to more indirect effects on photosynthesis and insect communication (Emmanuel de Langre, 2008).
Water
Water is probably the ‘element’ with the most legitimate claim to be part of a plant since it comprises up to 95% of the mass of growing plant tissues (Yoseph Negusse Araya & Gonzalo Garcia-Baquero, 2014; Karla Moeller & Charles Kazilek) and is the major component of the important cell organelle known as the vacuole (Chunhua Zhang et al., 2014). But water is not just a feature of the vacuole, it is an integral part of, or associated with, many macromolecules (Peter L Privalov & Colyn Crane-Robinson, 2017) and consequently is found throughout the cytoplasm, within organelles, and in relatively large amounts in the cell walls (Paul White et al., 2014; L Andrew Staehelin). However, it is the vacuole that is essential to growth of cells – and hence the whole plant – as it is the turgor of this organelle that drives cell expansion (Daniel J Cosgrove, 2014; Sabrina Kaiser & David Scheuring, 2020).
But water goes far beyond its role as a structural component of plants, it also fulfils many other offices: as a coolant (e.g., during transpiration – Susanne von Caemmerer & Neil Baker (2007)), as the driving force behind many growth movements (Yoël Forterre, 2013; J Edwards et al., 2019); as a translocation medium (e.g., for products of photosynthesis in the phloem – Hans Meidner & David Sheriff, 1976); as a reactant (e.g., in hydrolytic reactions – Hans Meidner & David Sheriff, 1976), and in photosynthesis; as a reaction medium (most plant chemistry takes place in an aqueous environment within cells – Hans Meidner & David Sheriff, 1976); as a germination-promoter (hydration of extremely dry tissues is the first stage in the germination process in most seeds – J Derek Bewley, 1997; Melissa Ha et al.; Nirmita Sharma, 2025); as a disperser of propagules (e.g., seed, fruit, fragments of mature plant, etc.), particularly for aquatic plants (Ajit Medhekan), hydrophytes (Nitu Borah); and as a ‘sexual facilitator’ (primarily for early-evolving plants with motile gametes such as ferns (Ernest M Gifford; Vannevar Bush [Ed. – and very occasionally in terrestrial flowering plants (Yong-Li Fan et al., 2019)]). Importantly, movement of water out of guard cells causes the stomata to close thereby reducing uncontrolled loss of water, which is particularly important in environments where water is in scant supply.
In fact, so important is water to plants that a whole suite of morphological, anatomical and physiological features have developed in environments: where it is scarce (e.g., reduced surface area-to-volume ratio of plants, thickened cuticles, spines (see Fig. at beginning of this chapter), succulence, and additional biochemistry such as CAM in some xerophytes (Harika Gupta; Maria Morrow)); where it is in abundance (hydrophytes (Maria Morrow)), or where water may be present but is too salty for most plants to be able to extract the life-saving water they need, as in the case of plants near the marine coast or on saline soils – so-called halophytes (Domingo M Jariel).
Under conditions of waterlogging (i.e., where there is considerable excess of water, but which is not present sufficiently long-term to be considered an aquatic environment), some plants undergo cell lysis which leads to development of cavities in the roots (LACJ Voesenek et al., 2006; Shunsaku Nishiuchi et al., 2012). This is a subtle response that relies upon the build-up of ethylene – a plant hormone (Melissa Ha et al.). Ethylene is naturally produced by the roots in small amounts, and usually diffuses away under normal conditions. However, under water-logging, the ethylene doesn’t readily diffuse away and its build-up within the plant leads to the production of the enzyme cellulase. Cellulase acts upon its substrate – cellulose – causing cell walls to breakdown and ultimately leads to the destruction of the cells. The lysigenous cavities so formed provide spaces for movement of gases – particularly oxygen – from the aerial to the submerged parts of the plant, such as the roots. Although it should be appreciated that the plant is not responding to excess water itself but rather to the low oxygen levels – hypoxia – that accompany the water-logging (Md. Alamgir Hossain & Sarder Nasir Uddin, 2011), either way, a Classical Element, whether Air or Water – is intimately involved in this behaviour.
And, in an evolutionary sense, if, as is widely acknowledged, life on Earth developed in an aquatic environment (Benton Clark & Vera Kolb, 2020), then water can be viewed as giving birth to the land flora since this was the habitat from which the ancestors of terrestrial plants evolved (Jan de Vries & John M Archibald, 2018; Mitchell Cruzan, 2019; ).
Getting the balance right
Just as ancient humoral medical practice believed that an imbalance of these four elements may cause disease in humans (Faith Lagay; Judy Duchan; Maya Prabhu; William Jackson, 2001), plants also experience dis-ease. Generally, the right balance of everything is required for a properly functioning individual. If one – or more – is in excess, e.g., flooding, or insufficiency of water as in arid deserts, plants will suffer in the short term. Which is a problem because environmental stresses are amongst the most serious constraints on current and future agriculture and global food security (Josie Garthwaite; Ron Mittler et al., 2025). Although, and seemingly counter-intuitively, irrigation of crops – to supply essential water in areas where it may be limited – often creates problems of salinisation, which can lead to significant plant stress and distress (Rana Munns & Matthew Gilliham, 2015; Kousik Atta et al., 2023). Yet, plants often find ways to cope with challenging environmental factors, and in the longer term they may become adapted to the prevailing conditions (i.e., they evolve).
Interconnectedness
And, in their turn, plants intimately interact with and affect these four ‘elements’ – for example they ‘purify’ air (Dong Sik Yang et al., 2009); they ameliorate the soil via their own natural phytoremediation activities (and exchange with the rhizosphere – Gilberto Curlango-Rivera et al., 2016); and they alter the quality and quantity of light that passes through their tissues – e.g., ratios of red/far-red light (Keera A Franklin & James R Shinkle, 2009). Furthermore, the escape of transpirational water [taken up by the plant from the soil] as vapour from their aerial surfaces into the atmosphere not only helps to cool the plant (Susanne von Caemmerer & Neil Baker, 2007), but also contributes to the creation of clouds and thereby impacts upon climate that affects the whole planet (Bonan, 2008). And in turn, plants are affected by global climate changes via changed patterns of rainfall, wind patterns, and temperature variations (Guy F Midgley, 2007).
Conclusion
This chapter emphasises the relative simplicity of the fundamental ‘ingredients’ – minerals from the soil, light and heat from the sun, oxygen and carbon dioxide from the atmosphere, and water – that are needed to create and sustain plant life. In this way, a modern-day reinterpretation of the Classical elements of earth, fire, air and water shows how that quartet of abiotic factors still helps to ‘create’ plants in the 21st century.
#AbioticFactors #Air #ClassicalElements #Earth #environmentalStresses #essentialElements #Fire #foodSecurity #germination #halophytes #hydrophytes #hypoxia #macronutrients #micronutrients #MineralNutrition #minerals #oxygen #photosynthesis #Rainfall #salinity #soil #sun #Temperature #water #WaterLogging #Wind #xerophytesAre #Wormholes Real? : Medium
#Scientists find #ClimateChange is reducing #Oxygen in #Rivers #Worldwide : Misc
The First #AtomicBomb Test in 1945 Created an Entirely New #Material : WIRED
Latest #KnowledgeLinks
https://atlas.whatip.xyz/post.php?slug=is-extracting-oxygen-from-lunar-soil-the-future-of-space-exploration
<p>As lunar missions are back in vogue with engineering more sustainable human presence on the Moon in mind
#exploration #extracting #missions #oxygen
✨🔬 For a long time we knew #gold stayed shiny because it resisted #oxygen.
Researchers at #Tulane University recently mapped the #physics behind this trait, and they found that surface #atoms on the metal rearrange themselves into a hexagonal structure that blocks #oxygen molecules from reacting.
👉 https://gizmodo.com/scientists-identify-atomic-trick-that-keeps-gold-shiny-2000761911
#chemistry #science #metallurgy #jewelry #elements #research #metals #education