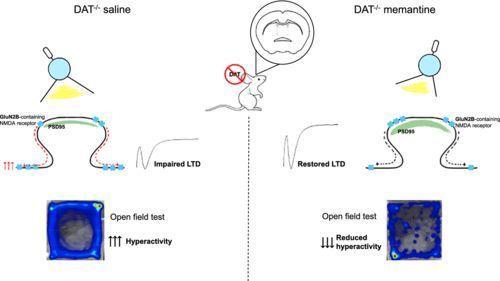
The article discusses a study that uses non-invasive brain scanning combined with computer modeling to measure how the Alzheimer’s drug memantine interacts with brain receptors in living patients, aiming to confirm drug action without invasive procedures. It also shows how these methods can track changes in brain activity over time in people with cognitive impairment or Alzheimer’s disease.
This approach is of interest to psychology readers because it demonstrates how brain activity and receptor dynamics relate to cognitive function and disease progression, and it highlights innovative methods for studying brain mechanisms in living humans.
Article Title: New brain scanning method safely tracks how Alzheimer’s drugs work in living patients
Link to PsyPost Article: https://www.psypost dot org/new-brain-scanning-method-safely-tracks-how-alzheimers-drugs-work-in-living-patients/
Copy and paste broken link above into your browser and replace "dot" with "." for link to work. We have to do it this way to avoid displaying copyrighted images.
#neuroimaging #alzheimersdisease #memantine #nmda-receptors #magnetoencephalography