Organocatalytic beta-glycosylation via SN1 type oxocarbenium: Benjamin List and coworkers from MPI developed a method to synthesize beta linked sugars from the trichloroacetimidate precursor by deploying a chiral acidic imidodiphosphorimidate catalyst. This work has been published in JACS 2025.
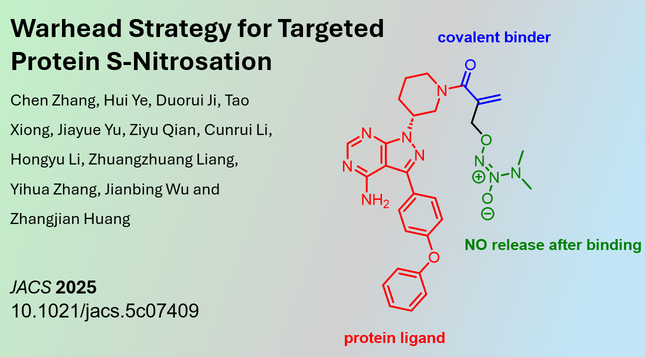
A novel bifunctional molecule induces selective nitrosation of BTK. Mechanistically, it first covalent bind to BTK and the release NO, which yielded in the nitrosation of a cysteine side chain. Published in #JACS by Jiangbing Wu, Zhangjian Huang and coworkers (#China)
Read more on doi: 10.1021/jacs.5c07409 or on my page: https://me.organicchemistry.eu/post/nitrosation-jacs-2025/
When you think of a reaction between an acid and an alcohol, you think of an ester as the product. MacMillan and co-workers from Princeton have now described a different pathway: Selective decarbonylative C-C coupling via a metal-catalyzed photoredox reaction. Read more about this in JACS:
Novel enantioselective #synthesis of rubriflordilactone B by Gui and colleagues from the Chinese Academy of Science published in #JACS
This is already the second total synthesis of this group, with an interesting reaction cascade as the key reaction in this process.
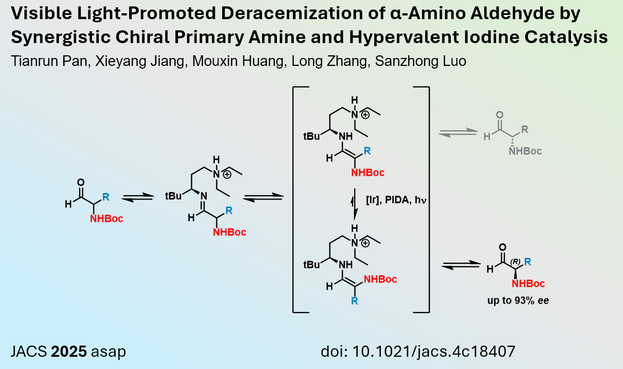
Luo et al. from Tsinghua University (#China) report in #JACS a novel method to create enantioenriched α-amino aldehydes using a chiral amine catalyst and iridium photocatalysis. Through this strategy α-amino aldehydes with up to 93% ee could be synthesised and subsequently stabilised via analogues formation (e.g. through Wittig reaction).
https://me.organicchemistry.eu/post/deracemization-aldehyde-jacs-2025/
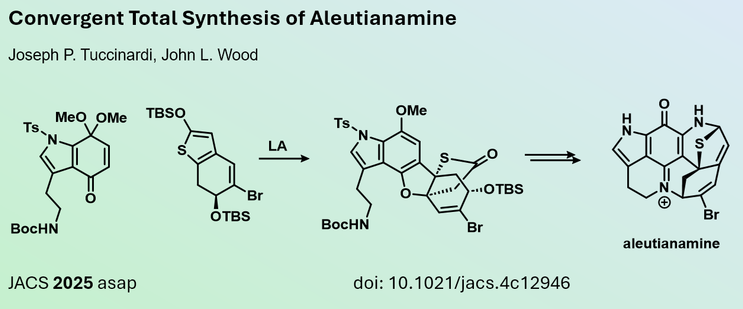
Joseph Tuccinardi and John Wood from Baylor University (#USA), introduced a novel strategy to efficiently synthesize the core scaffold of aleutianamine. Their study involved a vinylogous Mukaiyama-Michael reaction cascade using two previously unknown coupling partners. Published in #JACS
https://me.organicchemistry.eu/post/aleutianamine-jacs-2025/
Another total synthesis in the spotlight: Already early this year Mingji Dai and his research team described the total synthesis of heilonine in #JACS (doi: 10.1021/jacs.3c13492). This synthesis is short and elegant, using multiple C-H functionalisations and a Nazarov cyclisation for their key step.
Finaalllyyyy.... The first 𝐉𝐀𝐂𝐒 (IF - 14.5)😍🤩...
Special thanks to Felipe Fantuzzi (need any introduction? 3rd paper together🫂) & Holger group (University of Würzburg), Sougata Sarkar Sir (without whom I'd not even have an M.Sc.🙏)... & my mother (for insisting on asking Felipe to start a collaboration)😘😍
Link - https://pubs.acs.org/doi/10.1021/jacs.4c10967
#ACS #jacs #organometallic #computational #collaboration #journal #article #openaccess
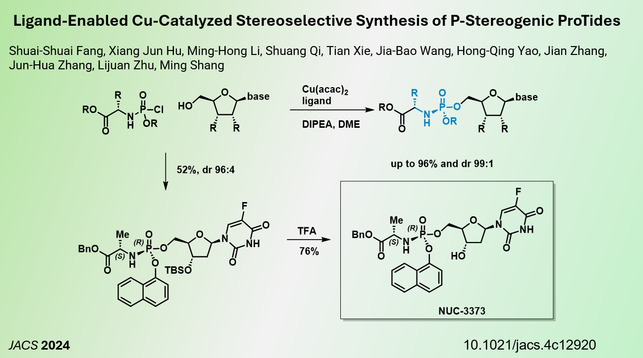
Efficient Synthesis of ProTide antivirals: A team from from #Fudan University and #Shanghai Jiao Tong University reporting a copper-catalyzed method for synthesizing chiral phosphates in #JACS.
This research represents a new tool for the synthesis of ProTide nucleoside analogues, providing an efficient route that can enhance the development of antiviral drugs in future studies.