One molecule could reverse aging, boost memory, and repair muscle
Imagine a pill that could slow aging, sharpen your memory, strengthen muscles, and reduce chronic inflammation. It sounds like science fiction, but scientists have discovered a molecule that could make this dream a reality. This molecule activates TERT, a key enzyme that protects DNA as we age. In lab tests, it didn’t just slow aging—it reversed many signs of it.
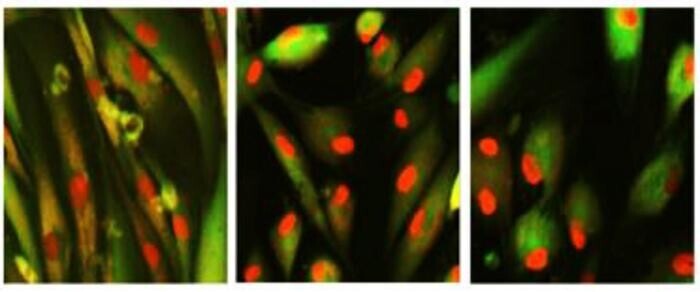
In studies with older models, six months of treatment with this TERT-activating compound (TAC) triggered new brain cell growth, improved genes related to memory, and reduced inflammation. It also cleared out damaged “zombie” cells, which are linked to aging, and restored muscle strength and coordination. In human cells, TAC helped cells divide longer by lowering DNA damage, essentially promoting healthier aging.
This breakthrough holds massive potential for age-related diseases like Alzheimer’s, Parkinson’s, heart disease, and cancer. While it’s still in the early stages, the possibility of reversing aging itself could soon be within our grasp.
The idea of stopping or even reversing the aging process is no longer just a fantasy. Thanks to this discovery, the future of medicine might be closer than we think.
Longevity & anti-aging
#Longevity #HealthyAging #ReversingAging #LifeExtension #AgeReversal
Molecular & cellular science
#TERT #Telomerase #CellularSenescence #MolecularBiology #DNARepair
Brain & muscle health
#Neurogenesis #BrainHealth #MuscleRegeneration #CognitiveHealth
Innovation & medicine
#MedicalBreakthrough #Biotech #RegenerativeMedicine #FutureOfMedicine