Very excited that Jon Stokes will visit us on May 28th for a @LED3hub Lecture. Very much looking forward to learning more about the potential of AI to speed up antibiotic discovery.
LED3hub
- 32 Followers
- 6 Following
- 341 Posts
We are looking forward to the LED3 Lecture by Jon Stokes on May 28th. He will talk about "An honest discussion about AI for antibiotic discovery "
If you are interested, make sure to come by!
https://www.universiteitleiden.nl/en/science/led3/led3-lectures
Great @LED3hub Lecture by Peng Chen. He talked about genetic code expansion in combination with in-cell chemistry to understand biology and develop therapeutics. Great insights into studying protein function with spatiotemporal resolution. (1/3)
We are very happy that Peng Chen from Peking University visited us today for a very well attended LED3 Lecture. He talked about "Live-cell Protein Chemistry in Health and Disease".
Very much looking forward to today's @LED3hub Lecture by Peng Chen on "Live-cell Protein Chemistry in Health and Disease".
If you are interested, make sure to come by!
https://www.universiteitleiden.nl/en/science/led3/led3-lectures
Do you want to hear the exciting and insightful stories behind drug discovery?
Lately, Stan Van Boeckel, emeritus professor at @leidenscience.bsky.social and @led3hub.bsky.social , has started a series of #SciComm posts on this topic. (1/4)
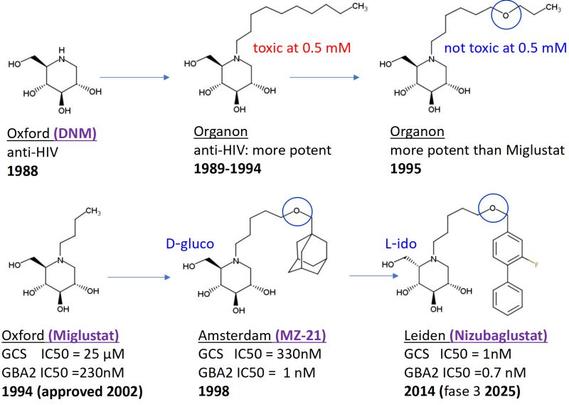
Another example—this time from my own history—of incremental improvement in drug discovery. In 1988 it was discovered in Oxford that deoxynojirimycin (DNM), an α‑glucosidase inhibitor found in… | Stan Van Boeckel
Another example—this time from my own history—of incremental improvement in drug discovery. In 1988 it was discovered in Oxford that deoxynojirimycin (DNM), an α‑glucosidase inhibitor found in mulberry leaves, inhibits HIV. At Organon I started a follow‑up program and we found that activity increased importantly with long N‑alkyl substituents on DNM. Unfortunately, those compounds were (cell)‑toxic due to strong amphiphilicity; however, inserting an oxygen atom into the long chain preserved activity while eliminating toxicity (a related molecule, UV‑4, recently reached Phase 1 as a broad‑spectrum antiviral). In Oxford they continued with shorter alkyl chains, which in 1994 laid the foundation for miglustat, approved in 2002 for lysosomal storage disorders through inhibition of GCS and GBA2. Overkleeft in Amsterdam returned in 1998 to the concept of a lipophilic side chain containing oxygen and created the more potent MZ21. Miglustat and MZ21, however, still inhibit α‑glucosidases in the GI tract, causing unpleasant side effects. The undesired α‑glucosidase activity could be minimized by giving the aza‑sugar the L‑Ido instead of the D‑Gluco configuration. Ultimately, the Overkleeft group and I (Leiden) combined all knowledge into nizubaglustat: highly (equi)potent on GCS and GBA2, CNS‑penetrant, and suitable for low once‑daily dosing. Nizubaglustat is now in Phase 3 clinical trials (NPC and Gangliosidosis by Azafaros) and will hopefully be approved before 2028—forty years after the first results with deoxynojirimycin (DNM).
He covers a variety of drug discovery stories with insightful science and a lot of interesting background.
Make sure to check them out and follow him, if you are interested. (2/4)
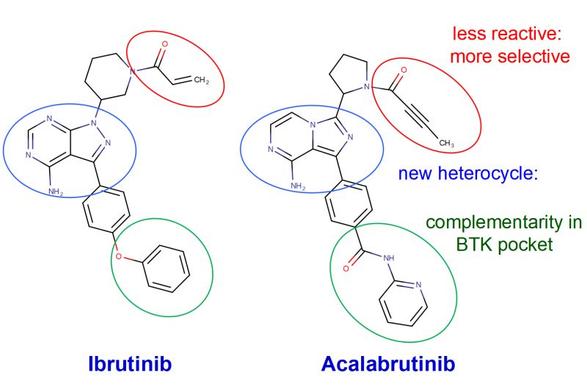
How did the Medicinal Chemists in my department at Organon create the new chemical entity that is now marketed as acalabrutinib (Calquence), a blockbuster BTK inhibitor? Initially, there was no BTK… | Stan Van Boeckel | 10 comments
How did the Medicinal Chemists in my department at Organon create the new chemical entity that is now marketed as acalabrutinib (Calquence), a blockbuster BTK inhibitor? Initially, there was no BTK project at all, only an Lck program. In that Lck project we were studying Src‑family selectivity, and in the screening panel a few compounds unexpectedly showed higher inhibitory activity against BTK. Several of the structural elements of those early leads can still be recognized in acalabrutinib. Jac Wijkmans urged me to continue developing these BTK inhibitors “under the radar” until they were strong enough to justify approval of a new project. Although the compounds were already reasonably potent and selective, I was inspired by a lecture from Daniel Rauh at a consortium meeting organized by Herbert Waldmann. Rauh explained the paradox that, for a small subset of kinases with an reactive cysteine in the ATP-binding pocket (e.g. EGFR, BTK, ITK), covalent binding to that cysteine can actually increase potency and selectivity. I pushed the team to pursue this covalent approach and, where possible, to install a mild Michael acceptor, since the inhibitors already had good complementarity in the BTK pocket (goal: better EGFR selectivity). Thus, the BTK project was not an attempt to “bust” the ibrutinib patent, but just a follow up of "the unexpected BTK leads". By the time acalabrutinib was later developed at Acerta and AstraZeneca, ibrutinib had already demonstrated clinical success in treating B‑cell malignancies. Indeed acalabrutinib shows better selectivity/safety in the clinic than ibrutinib. | 10 comments on LinkedIn
Other examples:
(3/4)
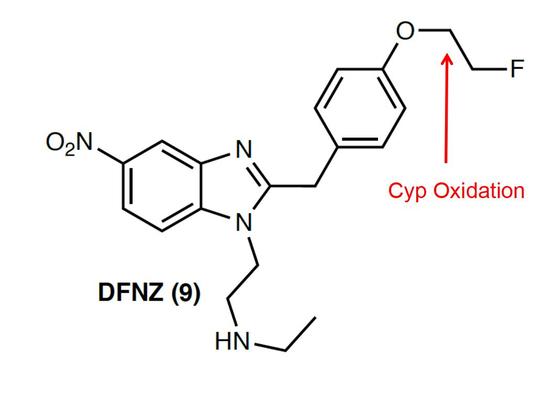
In this Nature article https://lnkd.in/eQsGRRvC they report N-destethylfluoronitazene (DFNZ) as a promising opioid receptor superagonist analgesic with minimal adverse effects (in Mice). Mind you… | Stan Van Boeckel | 15 comments
In this Nature article https://lnkd.in/eQsGRRvC they report N-destethylfluoronitazene (DFNZ) as a promising opioid receptor superagonist analgesic with minimal adverse effects (in Mice). Mind you, the O-fluoroethyl group is a huge liability as likely it will be cleaved by Cyp metabolism delivering highly toxic fluor-acetaldehyde and fluoro-acetic acid (inhibiting the Krebs cycle 💀 😱 ). Medicinal Chemists should avoid O(CH2CH2)n-F groups in their molecules . | 15 comments on LinkedIn
Are you a #PhD or #Postdoc and do you want to present your #ChemBio research to a wide international audience?
Here is your chance! Enter our Abstract Challenge to win a Flash Talk at the 6th Virtual ChemBioTalks on September 29th.
https://chemistry-europe.onlinelibrary.wiley.com/hub/journal/14397633/chembio-flash
If you do not want to miss any updates on the event, register for our mailing list here: https://fd24.formdesk.com/universiteitleiden/VirtualChemBioTalks







