
Identification of Bacterial FtsZ Effectors Targeting the Sites of Coumarin Binding - Cytology and Genetics
Abstract— There is a large group of bacterial FtsZ inhibitors, the biological activity of which has been confirmed biochemically. However, the sites of protein-ligand interaction for most of them remain unknown, significantly complicating the further search and combinatorial design of FtsZ inhibitors. This study presents the results of bioinformatic analysis of bacterial FtsZ effectors targeting sites of 4-hydroxycoumarin binding (BP1 and BP2). New data based on original results of pharmacophore screening, chemoinformatics, molecular docking, molecular dynamics simulations, AI-predictions, etc., are presented. The object of the study was a combined library of 379 compounds, formed based on revision of the structural database RCSB Protein Data Bank and biochemically proven FtsZ effectors from ChEMBL. Based on the results of a comprehensive study, 39 compounds were selected, of which 28 were identified as effectors of the sites BP1 and BP2, and another 11 as specific effectors of the site BP2 located in the BP2/IDC superpocket.

Chiral Isothiourea-Catalyzed Asymmetric (3+3) Cycloaddition - ChemistryViews
Enantioselective path to fluoroalkyl-substituted caprolactone-fused coumarins
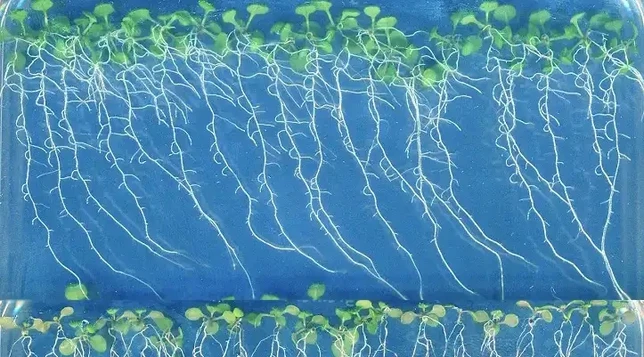
Iron is an essential micronutrient for plants. However, its availability to plants is highly dependent on the pH value of the soil. When suffering from iron deficiency, certain plant species release coumarins. Which type of these small secondary metabolites is mainly released depends on the pH value of the soil.
https://globalplantcouncil.org/plants-recruit-distinct-chemical-activities-of-coumarins-under-different-soil-phs/ via IPK Gatersleben #PlantScience #Science #Plants #Arabidopsis #Coumarins #agriculture #crops

Plants recruit distinct chemical activities of coumarins under different soil pHs - The Global Plant Council
Iron is an essential micronutrient for plants and is generally found in large quantities in the Earth's crust. However, its availability to plants is highly dependent on the pH value of the soil. When suffering from iron deficiency, certain plant species release coumarins. Which type of these small secondary metabolites is mainly released depends on the pH value of the soil. Now, a research team has shown how different coumarins are used to maximize iron uptake under acidic and alkaline conditions.

Plants found to recruit distinct chemical activities of coumarins under different soil pHs
Plants have two main uptake mechanisms to obtain iron (Fe) from the soils. The type of strategy employed depends on the botanical classification of the plant. In the so-called strategy-I mechanism, plants must first reduce the trivalent iron (Fe3+) into bivalent iron (Fe2+). Only then can Fe2+ be taken up by roots as a free ion.

Fluorescent blue coumarins in a folk-medicine plant could help us see inside cells
Plants that glow under ultraviolet (UV) light aren't just inventions of science fiction TV and movies. Roots of a traditional medicine plant called the orange climber, or Toddalia asiatica, can fluoresce an ethereal blue hue. And now, researchers have identified two coumarin molecules that could be responsible. These natural coumarins have unique fluorescent properties, and one of the compounds could someday be used for medical imaging. Their study is published in ACS Central Science.

