https://www.biorxiv.org/content/10.64898/2026.05.21.726871v1?rss=1
#Drosophila #Metabolism #Adult
#Larva
mühsam ernährt sich das eichhörnchen,
akrobatisch ernährt sich die raupe
der
Grasglucke
Euthrix potatoria
https://de.wikipedia.org/wiki/Grasglucke
#insekten #insects #krabbeltierchen #AuwaldRunde #BugOfTheDay #caterpillar #larva #nature #biodiversity #entomology
I've been going through around 5000 #iNaturalist plume #moth records in #Australia that are not yet research grade and for which I'd not yet added identifications. Almost all the species here can be identified well from a good photo.
The most interesting discovery today was a handful of #caterpillar observations that clearly represent the first photos of the #larva of the common species Stangeia xerodes. See the explanation here:
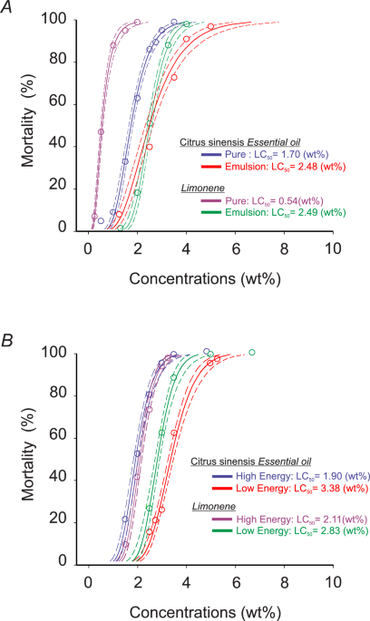
Sweet orange essential oil (Citrus sinensis) and its major component d-limonene were formulated as emulsions and low- and high-energy nanoemulsions and evaluated for toxicity against adults, larvae, and pupae of Drosophila suzukii, as well as for selectivity toward the pupal parasitoid Pachycrepoideus vindemmiae. High-energy nanoemulsions produced smaller and more homogeneous droplets (221–289 nm; PDI 0.210–0.255) compared with low-energy nanoemulsions (617–796 nm; PDI 0.548–0.644), indicating improved dispersion and potential stability. Toxicity assays revealed that the pure essential oil and d-limonene were the most toxic to adult D. suzukii (LC50 = 1.7 wt% and 0.54 wt%, respectively), followed by high-energy nanoemulsions, emulsions, and low-energy nanoemulsions. Although pure compounds showed slightly higher toxicity, high-energy nanoemulsions maintained comparable efficacy, suggesting improved delivery without major loss of activity. Larval mortality did not exceed 40% across treatments, whereas pupae were markedly more susceptible. High-energy nanoemulsions of both orange essential oil and d-limonene produced the highest pupal mortality (around 85%), followed by low-energy nanoemulsions (72–77.5%) and emulsions (50–75%). In addition, emulsified and nanoemulsified formulations induced developmental abnormalities in emerged adults, with malformation rates reaching 30%. Selectivity assays demonstrated low toxicity of all formulations to adult P. vindemmiae, with mortality generally below 20% and a negligible reduction of parasitism activity in most treatments. Overall, the formulation method strongly influenced physicochemical properties and insecticidal performance. High-energy nanoemulsions enhanced efficacy, particularly against pupae, while maintaining low toxicity to a key parasitoid. These findings indicate that nanoformulated citrus essential oils represent promising selective and environmentally compatible tools for integrated management of D. suzukii.
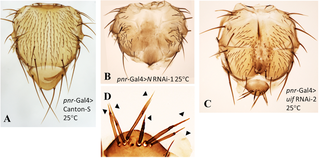
The Drosophila gene uninflatable (uif) encodes a conserved insect protein, first identified for its roles in the development and endopolyploid growth of several larval tissues. Uif is a transmembrane protein and its large extracellular domain contains several protein-protein interaction motifs, including multiple EGF (Epidermal Growth Factor-like) repeats. More recent studies have established that Uif can interact with Notch, a major regulator of Drosophila growth and differentiation, through its EGF repeats and have identified ways that Uif binding can modify Notch behavior. Endocytosis of Notch-Uif complexes into a particular class of endosomes has also been identified and implicated in cell fate decisions in the sense organs of the notum (thorax). We have examined uif’s functions in the Drosophila wing to assess possible roles in 1) growth of a mitotically derived tissue, 2) cell fate decisions in specialized wing structures, and 3) Notch-dependent processes. We used previously characterized Gal4 lines and RNAi constructs to suppress uif and Notch in different wing compartments. In addition to a role in mitotic growth throughout the wing, we have identified two new uif activities that are also shared with Notch: 1) regulation of pigment synthesis within the wing cuticle and 2) control of chemosensory sense organ number in the anterior wing margin through a role in an apoptosis-related mechanism. However, uif does not participate in two roles of Notch that regulate cell fate decisions: sense organ differentiation and formation of wing vein tissue. Given the similarities in the development of the notal and wing margin sense organs, we investigated further the previously proposed role for uif in differentiation of these structures on the notum. We found that loss of uif affects the growth of the bristles of the notal microchaete sense organs, but not their differentiation.