https://www.biorxiv.org/content/10.64898/2026.04.22.720250v1?rss=1
#Drosophila #Sleep
Open postdoc and PhD student positions in the lab of Carolina Rezaval (UK):
"We are recruiting three postdoctoral fellows and one research assistant/PhD student to join a Wellcome Trust-funded programme, ‘Decoding competition in the brain’."

Background IntS11, the core catalytic subunit of the Integrator (INT), functions in various stages of transcription. However, its specific function in orchestrating neural lineage progression remains poorly understood. Results Using a Drosophila model, we found that the absence of IntS11 leads to reduced brain size, which is not attributable to neuroblast (NB) loss or apoptosis but is due to impaired NB proliferation. MARCM (mosaic analysis with a repressible cell marker) revealed impaired clonal expansion, characterized by a significant decrease in progeny cell number. Live imaging and FUCCI analysis further confirmed a G1 arrest in IntS11-deficient NBs. Single-cell RNA-seq revealed downregulation of cell cycle regulators (aurB, CycE, Cdk4) in NBs and differentiation genes in GMCs, which were confirmed as candidates by ChIP-qPCR. Approximately 80% of neuronal morphogenesis–associated genes exhibiting 3′UTR shortening were downregulated. We further established that IntS11 physically binds to these loci to maintain lengthened 3′UTR isoforms and ensure mRNA stability in larval brains. Conclusions Collectively, our findings establish IntS11 as a stage-specific regulator of neural lineage progression that sustains NB proliferation through transcriptional control of cell-cycle genes and stabilizes differentiation programs by maintaining long 3′UTR isoforms.
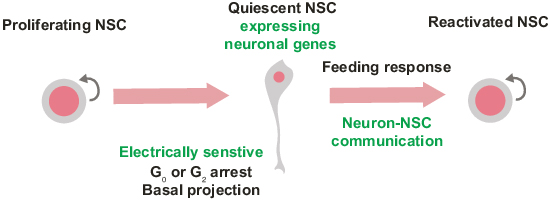
Reactivation of quiescent neural stem cells (NSCs) in the central nervous system (CNS) is a tightly controlled process that generates new neurons and glia to maintain homeostasis or enable repair post-injury, but it remains unclear if reactivation of distinct NSC populations is coupled. Here, we discovered that NSC quiescence exit in Drosophila follows a hierarchical sequence, whereby activation of anterior stem cells in the brain lobes precedes and is required for the timely state-transition of more posterior NSCs in the ventral nerve cord. To achieve this, quiescent NSCs transiently activate neuronal genes. This transient neuronal state is temporary and specific to NSC dormancy, as neuronal genes are switched off after stem cells resume proliferation. Blocking neuronal firing in brain lobe neurons delays the onset of posterior NSC reactivation. Our results reveal long-range communication between quiescent NSCs to coordinate reactivation across the CNS, enabled by a transient, plastic neuron-like state that allows direct interaction with neuronal axons.