#mwgic #2026 #Osteoarthritis #OA
#HumanTrials #FDAapproval
https://medicalxpress.com/news/2026-06-hydrogel-relieves-osteoarthritis-pain-cartilage.html
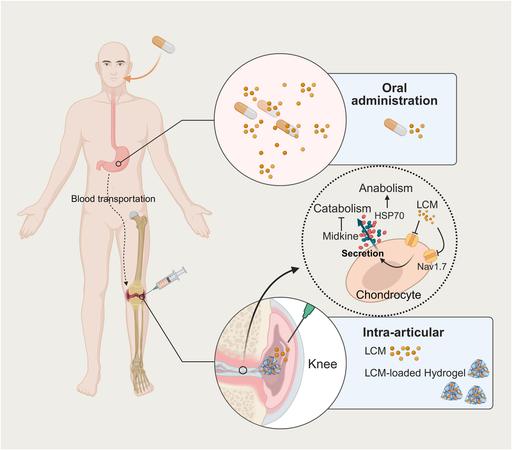
Injectable hydrogel relieves osteoarthritis pain and repairs cartilage in preclinical tests
For millions of people living with osteoarthritis, daily life can involve a frustrating cycle of pain and stiffness. While current treatments like over-the-counter medications or steroid injections can temporarily dull the ache, they do not stop the joint from deteriorating. A Yale study published in the journal Bioactive Materials found that the medication lacosamide acts as a highly effective, dual-purpose treatment that relieves joint pain and reverses cartilage damage in osteoarthritis, especially when a specialized hydrogel delivers the drug directly into the joint.