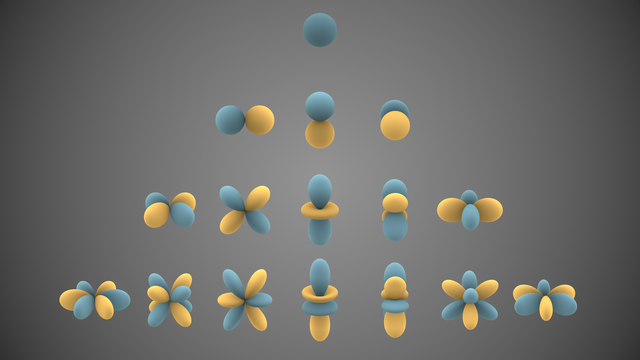@darthvader42 @ginger_tosser @TheBreadmonkey
Yes. But at least now you have a *rich and deep* lack of understanding of Quantum Mechanics.
😆
@ginger_tosser @TheBreadmonkey
But now you understand that you don't understand it.
It makes no sense but that’s just the way QM is and you just have to roll with it.
@swggrkllr3rd @ginger_tosser @freequaybuoy
I forgot that this film was scientifically accurate and based on factual events
If you think you understand Quantum Physics then you don't understand Quantum Physics.
But can he draw a perfect circle in one motion on that bad boy behind him?
God yes, he really is correct.
I briefly studied quantum mechanics when I had a toddler and a newborn baby and let me tell you, quantum mechanics and toddlers is a verybad combination.
Mind you, earlier on when I was doing some planetary science the toddler could recite Keplers Laws of Planetary Motion, mostly because I muttered them under my breath a lot to help me remember them. (I have since forgotten them)
@TheBreadmonkey what's actually non-intuitive about quantum mechanics? I suggest that whatever problems with "intuition" are getting in the way, are synthetic ones: people who are brought up with one set of arbitrary expectations about the physical world (i.e. that everything is, or ought to be, down to the deterministic behavior of discrete particles) are suddenly taught something else, and this produces the "non-intuitive" difficulties.
It's not that much different from chemistry is reckoned as confusing—because people are taught an arbitrarily simplified formalism at the start, and LATER get taught "oh actually things are more complicated than that". I think it amounts to deliberate obfuscation. The educational systems of "the West" are practically intended to confuse most people, in order to weed a select few geniuses—geniuses who are perceived to be geniuses along certain social and racial lines, mind you.
@mxchara Things like the bullshit we were taught in school: ”electrons circle like planets around the nucleus”, and then ”electrons jump to a higher shell when radiation hits them, you know those shells that you were taught in chemistry”, leading my 14 year old brain to think ”What? That doesn’t make any sense at all”.
I finally understood in my 30’s when I took a proper university physics course, that included quantum mechanics. The mystery of the 1.8V red LED:s also ceased to be a mystery.
@sleepybisexual @ahltorp hm. I've been trying to think of how to explain this myself.
Let me start with a much simpler physical system that also displays quantized behavior: a guitar string, constrained at both ends. The string vibrates back and forth, but the physical constraints of the system—the properties of the string itself and the distance between its endpoints—compel the string to vibrate only in certain modes, certain frequencies that we call the "fundamental mode" (the lowest possible frequency of vibration, in which the whole string is swinging back and forth in unison and only the endpoints are stationary) and its "overtones".
There's an infinite series of vibrational modes of the string, in theory, but in practice, if you were to measure the vibrations of the string over a long time and analyze them in the frequency domain, you'd find that most of the energy of the string's vibration went into the fundamental mode and the first few overtones.
These modes are equivalent to the excitational modes of electrons vibrating or oscillating around a nucleus.
(cont'd)
@sleepybisexual @ahltorp That system is more complicated because (a) it's three dimensional, and (b) at high enough energies of vibration, one must take special relativity into account, not something you'll usually worry about with guitar strings. All the same, the overall character of the physical system is roughly equivalent: there's a highly mobile thing that's free to wobble about (the electron cloud) and physical constraints (the force of attraction to the nucleus) which cause that cloud of electrons to be limited to moving about within a definite set of modes.
We call those modes "orbitals", because they're roughly analogous to the gravitational orbits of discrete bodies, but in fact orbitals are three-dimensional shapes and they're defined not as specific paths or lines which the electrons take, but as regions of probability density. one is more or less likely to find the electron at certain distances and solid angles around the nucleus, in these various orbitals.
(cont'd)
@sleepybisexual @ahltorp The excitational modes of electrons moving around a nucleus, because they're defined in three-dimensional space, are quantized according to what are called "spherical harmonics" (q.v. https://en.wikipedia.org/wiki/Spherical_harmonics) which turn up in other physical systems, such as the vibrations of the surface of a elastic sphere.
Now, back to the model of the guitar string. If you put more kinetic energy generally into a vibrating guitar string, it doesn't just vibrate faster or more. It puts more and more energy into the higher modes of vibration, higher overtones, which are at a higher "energy level" above the baseline energy of the string vibrating in its fundamental model. The same thing applies to electrons round nuclei. When they get more excited and energetic, they're still constrained to their sets of orbitals, but they can go into higher-energy orbitals further away from the nucleus (and therefore liable to become engaged in chemical reactions.)


while very funny, I find it a little off putting to think about thousands of millions of 18 yro paying millions of dollars to be told this. ya'll coulda just become electricians at $300 an hour and also a benefit to humanity
If humans enjoy doing it, it's a benefit to them. I like learning. That's not a waste of my time. That's something I want to do with my time. Value is personal.
It took me a long time to reconcile this point. I look at sports and have similar feelings about wasted time.
Jokes aside, I've never seen Heisenberg's Uncertainty Principle explained better than in the first lecture here!
If anybody is serious, be sure to check this one out!
@TheBreadmonkey
@rameshgupta @TheBreadmonkey Dagnabbit!
Just as #FreeTube broke (fixed and waiting for the new version to be released). OK, I need to update #ytdlp as well by the look of it.
Thanks for the tip though, looks like an interesting set of lectures.
I might let First Born watch this later.
@TheBreadmonkey Love it!
(PhD 9T6 University of Toronto Graduate Department of Physics)