
Post-doctoral researcher in oncology and computer science
Post a job in 3min, or find thousands of job offers like this one at jobRxiv!
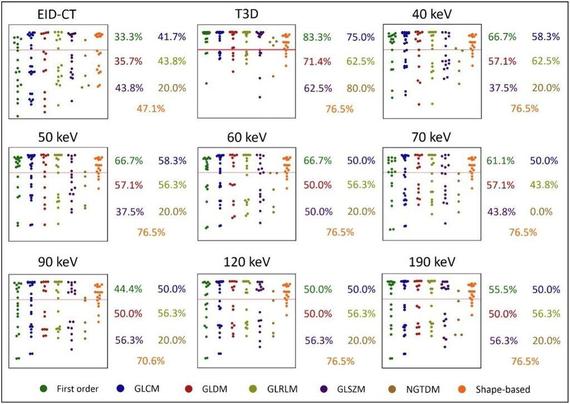
This #EuropeanRadiologyExperimental study by Giuseppe Tremamunno et al. evaluated the impact of novel photon-counting detector CT (PCD-CT) on the stability and interscanner/interreader reproducibility of myocardial #radiomic features of a cohort of patients undergoing CCTA on both energy-integrating detector CT (EID-CT) and PCD-CT.
🔗 https://buff.ly/40gGPHS

Intraindividual reproducibility of myocardial radiomic features between energy-integrating detector and photon-counting detector CT angiography - European Radiology Experimental
Background Radiomics is not yet used in clinical practice due to concerns regarding its susceptibility to technical factors. We aimed to assess the stability and interscan and interreader reproducibility of myocardial radiomic features between energy-integrating detector computed tomography (EID-CT) and photon-counting detector CT (PCD-CT) in patients undergoing coronary CT angiography (CCTA) on both systems. Methods Consecutive patients undergoing clinically indicated CCTA on an EID-CT were prospectively enrolled for a PCD-CT CCTA within 30 days. Virtual monoenergetic images (VMI) at various keV levels and polychromatic images (T3D) were generated for PCD-CT, with image reconstruction parameters standardized between scans. Two readers performed myocardial segmentation and 110 radiomic features were compared intraindividually between EID-CT and PDC-CT series. The agreement of parameters was assessed using the intraclass correlation coefficient and paired t-test for the stability of the parameters. Results Eighteen patients (15 males) aged 67.6 ± 9.7 years (mean ± standard deviation) were included. Besides polychromatic PCD-CT reconstructions, 60- and 70-keV VMIs showed the highest feature stability compared to EID-CT (96%, 90%, and 92%, respectively). The interscan reproducibility of features was moderate even in the most favorable comparisons (median ICC 0.50 [interquartile range 0.20–0.60] for T3D; 0.56 [0.33–0.74] for 60 keV; 0.50 [0.36–0.62] for 70 keV). Interreader reproducibility was excellent for the PCD-CT series and good for EID-CT segmentations. Conclusion Most myocardial radiomic features remain stable between EID-CT and PCD-CT. While features demonstrated moderate reproducibility between scanners, technological advances associated with PCD-CT may lead to greater reproducibility, potentially expediting future standardization efforts. Relevance statement While the use of PCD-CT may facilitate reduced interreader variability in radiomics analysis, the observed interscanner variations in comparison to EID-CT should be taken into account in future research, with efforts being made to minimize their impact in future radiomics studies. Key Points Most myocardial radiomic features resulted in being stable between EID-CT and PCD-CT on certain VMIs. The reproducibility of parameters between detector technologies was limited. PCD-CT improved interreader reproducibility of myocardial radiomic features. Graphical Abstract
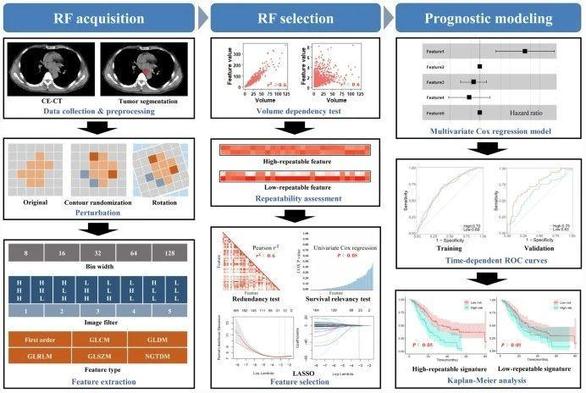
This study recently published in #InsightsIntoImaging investigated the repeatability of #radiomic features and its impact on the cross-institutional generalizability of the prognostic model for predicting local recurrence-free survival and overall survival of esophageal squamous cell #cancer receiving definitive chemoradiotherapy. (Jie Gong et al.)
🔗 https://buff.ly/3Yl5qKx

Using high-repeatable radiomic features improves the cross-institutional generalization of prognostic model in esophageal squamous cell cancer receiving definitive chemoradiotherapy - Insights into Imaging
Objectives Repeatability is crucial for ensuring the generalizability and clinical utility of radiomics-based prognostic models. This study aims to investigate the repeatability of radiomic feature (RF) and its impact on the cross-institutional generalizability of the prognostic model for predicting local recurrence-free survival (LRFS) and overall survival (OS) in esophageal squamous cell cancer (ESCC) receiving definitive (chemo) radiotherapy (dCRT). Methods Nine hundred and twelve patients from two hospitals were included as training and external validation sets, respectively. Image perturbations were applied to contrast-enhanced computed tomography to generate perturbed images. Six thousand five hundred ten RFs from different feature types, bin widths, and filters were extracted from the original and perturbed images separately to evaluate RF repeatability by intraclass correlation coefficient (ICC). The high-repeatable and low-repeatable RF groups grouped by the median ICC were further analyzed separately by feature selection and multivariate Cox proportional hazards regression model for predicting LRFS and OS. Results First-order statistical features were more repeatable than texture features (median ICC: 0.70 vs 0.42–0.62). RFs from LoG had better repeatability than that of wavelet (median ICC: 0.70–0.84 vs 0.14–0.64). Features with smaller bin widths had higher repeatability (median ICC of 8–128: 0.65–0.47). For both LRFS and OS, the performance of the models based on high- and low-repeatable RFs remained stable in the training set with similar C-index (LRFS: 0.65 vs 0.67, p = 0.958; OS: 0.64 vs 0.65, p = 0.651), while the performance of the model based on the low-repeatable group was significantly lower than that based on the high-repeatable group in the external validation set (LRFS: 0.61 vs 0.67, p = 0.013; OS: 0.56 vs 0.63, p = 0.013). Conclusions Applying high-repeatable RFs in modeling could safeguard the cross-institutional generalizability of the prognostic model in ESCC. Critical relevance statement The exploration of repeatable RFs in different diseases and different types of imaging is conducive to promoting the proper use of radiomics in clinical research. Key Points The repeatability of RFs impacts the generalizability of the radiomic model. The high-repeatable RFs safeguard the cross-institutional generalizability of the model. Smaller bin width helps improve the repeatability of RFs. Graphical Abstract
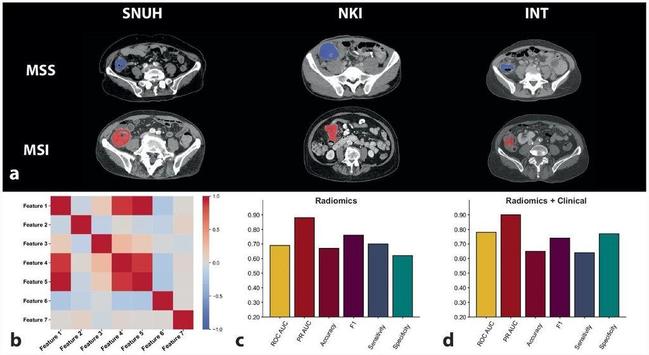
Zuhir Bodalal et al. looked at the association between tumor morphology and that status of microsatellite instability (MSI) vs microsatellite stability (MSS). They discovered that the differences in the #radiomic morphological phenotype between tumors MSI or MSS could be detected using radiogenomic approaches.
#EuropeanRadiologyExperimental
🔗 https://buff.ly/3BMXqsY

Non-invasive CT radiomic biomarkers predict microsatellite stability status in colorectal cancer: a multicenter validation study - European Radiology Experimental
Background Microsatellite instability (MSI) status is a strong predictor of response to immunotherapy of colorectal cancer. Radiogenomic approaches promise the ability to gain insight into the underlying tumor biology using non-invasive routine clinical images. This study investigates the association between tumor morphology and the status of MSI versus microsatellite stability (MSS), validating a novel radiomic signature on an external multicenter cohort. Methods Preoperative computed tomography scans with matched MSI status were retrospectively collected for 243 colorectal cancer patients from three hospitals: Seoul National University Hospital (SNUH); Netherlands Cancer Institute (NKI); and Fondazione IRCCS Istituto Nazionale dei Tumori, Milan Italy (INT). Radiologists delineated primary tumors in each scan, from which radiomic features were extracted. Machine learning models trained on SNUH data to identify MSI tumors underwent external validation using NKI and INT images. Performances were compared in terms of area under the receiving operating curve (AUROC). Results We identified a radiomic signature comprising seven radiomic features that were predictive of tumors with MSS or MSI (AUROC 0.69, 95% confidence interval [CI] 0.54−0.84, p = 0.018). Integrating radiomic and clinical data into an algorithm improved predictive performance to an AUROC of 0.78 (95% CI 0.60−0.91, p = 0.002) and enhanced the reliability of the predictions. Conclusion Differences in the radiomic morphological phenotype between tumors MSS or MSI could be detected using radiogenomic approaches. Future research involving large-scale multicenter prospective studies that combine various diagnostic data is necessary to refine and validate more robust, potentially tumor-agnostic MSI radiogenomic models. Relevance statement Noninvasive radiomic signatures derived from computed tomography scans can predict MSI in colorectal cancer, potentially augmenting traditional biopsy-based methods and enhancing personalized treatment strategies. Key Points Noninvasive CT-based radiomics predicted MSI in colorectal cancer, enhancing stratification. A seven-feature radiomic signature differentiated tumors with MSI from those with MSS in multicenter cohorts. Integrating radiomic and clinical data improved the algorithm’s predictive performance. Graphical Abstract
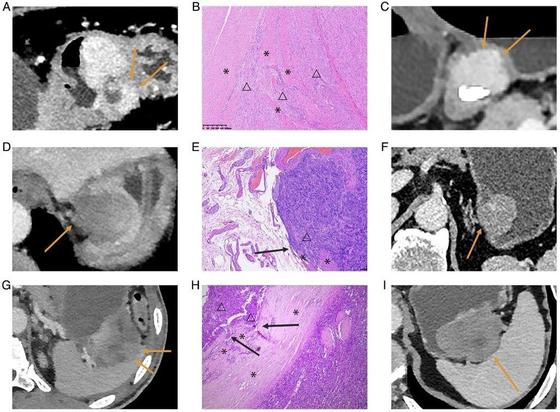
Xiaoxuan Jia et al. investigated the correlation of the mitotic index of gastric gastrointestinal stromal tumors with CT-identified morphological and first-order #radiomic features, finding that the invasive margin could be the sole independent CT high-risk morphological feature for 1–5 cm gGISTs after tumor size-based subgroup analysis.
#EuropeanRadiology
🔗 https://buff.ly/3NpjAnC

CT assessed morphological features can predict higher mitotic index in gastric gastrointestinal stromal tumors - European Radiology
Objectives To investigate the correlation of the mitotic index (MI) of 1–5 cm gastric gastrointestinal stromal tumors (gGISTs) with CT-identified morphological and first-order radiomics features, incorporating subgroup analysis based on tumor size. Methods We enrolled 344 patients across four institutions, each pathologically diagnosed with 1–5 cm gGISTs and undergoing preoperative contrast-enhanced CT scans. Univariate and multivariate analyses were performed to investigate the independent CT morphological high-risk features of MI. Lesions were categorized into four subgroups based on their pathological LD: 1–2 cm (n = 69), 2–3 cm (n = 96), 3–4 cm (n = 107), and 4–5 cm (n = 72). CT morphological high-risk features of MI were evaluated in each subgroup. In addition, first-order radiomics features were extracted on CT images of the venous phase, and the association between these features and MI was investigated. Results Tumor size (p = 0.04, odds ratio, 1.41; 95% confidence interval: 1.01–1.96) and invasive margin (p < 0.01, odds ratio, 4.55; 95% confidence interval: 1.77–11.73) emerged as independent high-risk features for MI > 5 of 1–5 cm gGISTs from multivariate analysis. In the subgroup analysis, the invasive margin was correlated with MI > 5 in 3–4 cm and 4–5 cm gGISTs (p = 0.02, p = 0.03), and potentially correlated with MI > 5 in 2–3 cm gGISTs (p = 0.07). The energy was the sole first-order radiomics feature significantly correlated with gGISTs of MI > 5, displaying a strong correlation with CT-detected tumor size (Pearson’s ρ = 0.85, p < 0.01). Conclusions The invasive margin stands out as the sole independent CT morphological high-risk feature for 1–5 cm gGISTs after tumor size-based subgroup analysis, overshadowing intratumoral morphological characteristics and first-order radiomics features. Key Points Question How can accurate preoperative risk stratification of gGISTs be achieved to support treatment decision-making? Findings Invasive margins may serve as a reliable marker for risk prediction in gGISTs up to 5 cm, rather than surface ulceration, irregular shape, necrosis, or heterogeneous enhancement. Clinical relevance For gGISTs measuring up to 5 cm, preoperative prediction of the metastatic risk could help select patients who could be treated by endoscopic resection, thereby avoiding overtreatment.
Rui Lv et al. investigated the relationship between #radiomic features in perivascular adipose tissue and abdominal aortic aneurysm growth following endovascular aneurysm repair.
#InsightsIntoImaging
🔗 https://buff.ly/4dCGceM
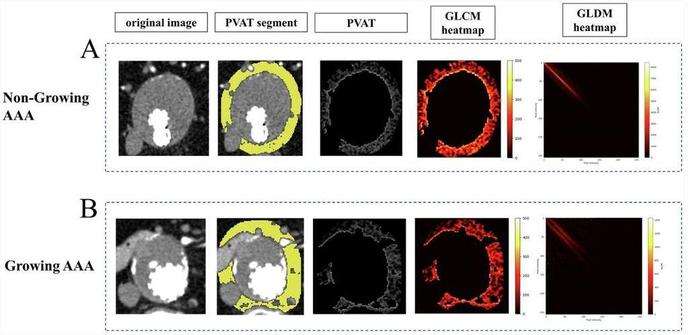

Assessing abdominal aortic aneurysm growth using radiomic features of perivascular adipose tissue after endovascular repair - Insights into Imaging
Objectives The study aimed to investigate the relationship between the radiomic features of perivascular adipose tissue (PVAT) and abdominal aortic aneurysm (AAA) growth after endovascular aneurysm repair (EVAR). Methods Patients with sub-renal AAA who underwent regular follow-up after EVAR between March 2014 and March 2024 were retrospectively collected. Two radiologists segmented aneurysms and PVAT. Patients were categorised into growing and non-growing groups based on volumetric changes observed in two follow-up computed tomography examinations. One hundred seven radiomic features were automatically extracted from the PVAT region. Univariable and multivariable logistic regression was performed to analyse radiomic features and clinical characteristics. Furthermore, the performance of the integrated clinico-radiological model was compared with models using only radiomic features or clinical characteristics separately. Results A total of 79 patients (68 ± 9 years, 89% men) were enroled in this study, 19 of whom had a growing aneurysm. Compared to the non-growing group, PVAT of growing AAA showed a higher surface area to volume ratio (non-growing vs growing, 0.63 vs 0.70, p = 0.04), and a trend of low dependence and high dispersion manifested by texture features (p < 0.05). The area under the curve of the integrated clinico-radiological model was 0.78 (95% confidence intervals 0.65–0.91), with a specificity of 87%. The integrated model outperformed models using only radiomic or clinical features separately (0.78 vs 0.69 vs 0.69). Conclusions Higher surface area to volume ratio and more heterogeneous texture presentation of PVAT were associated with aneurysm dilation after EVAR. Radiomic features of PVAT have the potential to predict AAA progression. Clinical relevance statement Radiomic features of PVAT are associated with AAA progression and can be an independent risk factor for aneurysm dilatation to assist clinicians in postoperative patient surveillance and management. Key Points After EVAR for AAA, patients require monitoring for progression. PVAT surrounding growing AAA after EVAR exhibits a more heterogeneous texture. Integrating PVAT-related features and clinical features results in better predictive performance. Graphical Abstract
In their "Shape Matters" #Radiomics study, Martha Foltyn-Dumitru et al. identify IDH-wild type glioma phenotypes and assess their impact on overall survival by clustering based on #radiomic features such as shape and tumor volume. Their technique could help improve the prediction of survival outcomes in high-grade glioma patients.
#EuropeanRadiology
🔗 https://buff.ly/4dhICPW
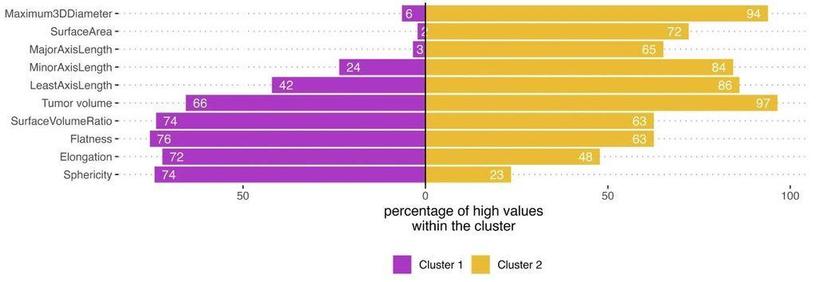

Shape matters: unsupervised exploration of IDH-wildtype glioma imaging survival predictors - European Radiology
Objectives This study examines clustering based on shape radiomic features and tumor volume to identify IDH-wildtype glioma phenotypes and assess their impact on overall survival (OS). Materials and methods This retrospective study included 436 consecutive patients diagnosed with IDH-wt glioma who underwent preoperative MR imaging. Alongside the total tumor volume, nine distinct shape radiomic features were extracted using the PyRadiomics framework. Different imaging phenotypes were identified using partition around medoids (PAM) clustering on the training dataset (348/436). The prognostic efficacy of these phenotypes in predicting OS was evaluated on the test dataset (88/436). External validation was performed using the public UCSF glioma dataset (n = 397). A decision-tree algorithm was employed to determine the relevance of features associated with cluster affiliation. Results PAM clustering identified two clusters in the training dataset: Cluster 1 (n = 233) had a higher proportion of patients with higher sphericity and elongation, while Cluster 2 (n = 115) had a higher proportion of patients with higher maximum 3D diameter, surface area, axis lengths, and tumor volume (p < 0.001 for each). OS differed significantly between clusters: Cluster 1 showed a median OS of 23.8 compared to 11.4 months of Cluster 2 in the holdout test dataset (p = 0.002). Multivariate Cox regression showed improved performance with cluster affiliation over clinical data alone (C index 0.67 vs 0.59, p = 0.003). Cluster-based models outperformed the models with tumor volume alone (evidence ratio: 5.16–5.37). Conclusion Data-driven clustering reveals imaging phenotypes, highlighting the improved prognostic power of combining shape-radiomics with tumor volume, thereby outperforming predictions based on tumor volume alone in high-grade glioma survival outcomes. Clinical relevance statement Shape-radiomics and volume-based cluster analyses of preoperative MRI scans can reveal imaging phenotypes that improve the prediction of OS in patients with IDH-wild type gliomas, outperforming currently known models based on tumor size alone or clinical parameters. Key Points Shape radiomics and tumor volume clustering in IDH-wildtype gliomas are investigated for enhanced prognostic accuracy. Two distinct phenotypic clusters were identified with different median OSs. Integrating shape radiomics and volume-based clustering enhances OS prediction in IDH-wildtype glioma patients.
Hong Wei et al. investigate the utility of #DeepLearning automated segmentation-based MRI #radiomic features to predict the early recurrence of single hepatocellular carcinoma (#HCC).
#InsightsIntoImaging
🔗 https://buff.ly/4bs961a
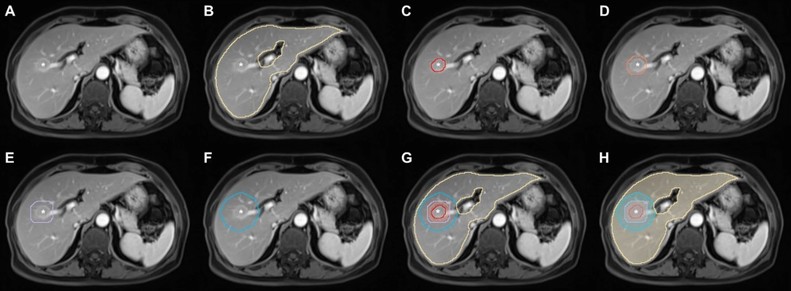

MRI radiomics based on deep learning automated segmentation to predict early recurrence of hepatocellular carcinoma - Insights into Imaging
Objectives To investigate the utility of deep learning (DL) automated segmentation-based MRI radiomic features and clinical-radiological characteristics in predicting early recurrence after curative resection of single hepatocellular carcinoma (HCC). Methods This single-center, retrospective study included consecutive patients with surgically proven HCC who underwent contrast-enhanced MRI before curative hepatectomy from December 2009 to December 2021. Using 3D U-net-based DL algorithms, automated segmentation of the liver and HCC was performed on six MRI sequences. Radiomic features were extracted from the tumor, tumor border extensions (5 mm, 10 mm, and 20 mm), and the liver. A hybrid model incorporating the optimal radiomic signature and preoperative clinical-radiological characteristics was constructed via Cox regression analyses for early recurrence. Model discrimination was characterized with C-index and time-dependent area under the receiver operating curve (tdAUC) and compared with the widely-adopted BCLC and CNLC staging systems. Results Four hundred and thirty-four patients (median age, 52.0 years; 376 men) were included. Among all radiomic signatures, HCC with 5 mm tumor border extension and liver showed the optimal predictive performance (training set C-index, 0.696). By incorporating this radiomic signature, rim arterial phase hyperenhancement (APHE), and incomplete tumor “capsule,” a hybrid model demonstrated a validation set C-index of 0.706 and superior 2-year tdAUC (0.743) than both the BCLC (0.550; p < 0.001) and CNLC (0.635; p = 0.032) systems. This model stratified patients into two prognostically distinct risk strata (both datasets p < 0.001). Conclusion A preoperative imaging model incorporating the DL automated segmentation-based radiomic signature with rim APHE and incomplete tumor “capsule” accurately predicted early postsurgical recurrence of a single HCC. Critical relevance statement The DL automated segmentation-based MRI radiomic model with rim APHE and incomplete tumor “capsule” hold the potential to facilitate individualized risk estimation of postsurgical early recurrence in a single HCC. Key Points A hybrid model integrating MRI radiomic signature was constructed for early recurrence prediction of HCC. The hybrid model demonstrated superior 2-year AUC than the BCLC and CNLC systems. The model categorized the low-risk HCC group carried longer RFS. Graphical Abstract
New #Radiomics in GU Imaging study. Annemarie Uhlig et al. discover that #radiomic feature analyses is able to distinguish renal tumor subtypes on routinely acquired CTs.
#EuropeanRadiology
🔗 https://buff.ly/4aYovFX
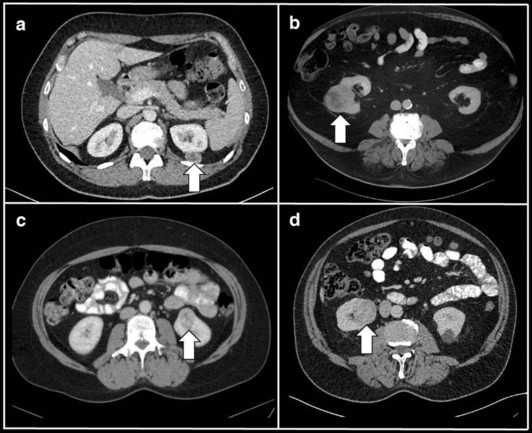

Radiomics and machine learning for renal tumor subtype assessment using multiphase computed tomography in a multicenter setting - European Radiology
Objectives To distinguish histological subtypes of renal tumors using radiomic features and machine learning (ML) based on multiphase computed tomography (CT). Material and methods Patients who underwent surgical treatment for renal tumors at two tertiary centers from 2012 to 2022 were included retrospectively. Preoperative arterial (corticomedullary) and venous (nephrogenic) phase CT scans from these centers, as well as from external imaging facilities, were manually segmented, and standardized radiomic features were extracted. Following preprocessing and addressing the class imbalance, a ML algorithm based on extreme gradient boosting trees (XGB) was employed to predict renal tumor subtypes using 10-fold cross-validation. The evaluation was conducted using the multiclass area under the receiver operating characteristic curve (AUC). Algorithms were trained on data from one center and independently tested on data from the other center. Results The training cohort comprised n = 297 patients (64.3% clear cell renal cell cancer [RCC], 13.5% papillary renal cell carcinoma (pRCC), 7.4% chromophobe RCC, 9.4% oncocytomas, and 5.4% angiomyolipomas (AML)), and the testing cohort n = 121 patients (56.2%/16.5%/3.3%/21.5%/2.5%). The XGB algorithm demonstrated a diagnostic performance of AUC = 0.81/0.64/0.8 for venous/arterial/combined contrast phase CT in the training cohort, and AUC = 0.75/0.67/0.75 in the independent testing cohort. In pairwise comparisons, the lowest diagnostic accuracy was evident for the identification of oncocytomas (AUC = 0.57–0.69), and the highest for the identification of AMLs (AUC = 0.9–0.94) Conclusion Radiomic feature analyses can distinguish renal tumor subtypes on routinely acquired CTs, with oncocytomas being the hardest subtype to identify. Clinical relevance statement Radiomic feature analyses yield robust results for renal tumor assessment on routine CTs. Although radiologists routinely rely on arterial phase CT for renal tumor assessment and operative planning, radiomic features derived from arterial phase did not improve the accuracy of renal tumor subtype identification in our cohort.
In this study recently published in #InsightsIntoImaging, Hai-Feng Liu et al. highlight the potential of a multiparametric MRI-based #radiomic model that integrates intratumoral and peritumoral features for predicting #HCC differentiation.
🔗 https://buff.ly/3U5HOqX
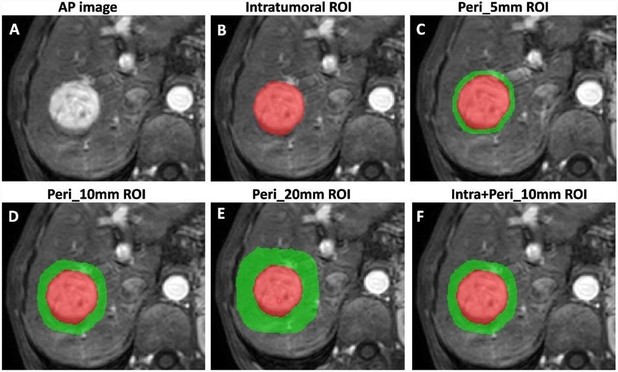

Multiparametric MRI-based intratumoral and peritumoral radiomics for predicting the pathological differentiation of hepatocellular carcinoma - Insights into Imaging
Purpose To explore the predictive potential of intratumoral and multiregion peritumoral radiomics features extracted from multiparametric MRI for predicting pathological differentiation in hepatocellular carcinoma (HCC) patients. Methods A total of 265 patients with 277 HCCs (training cohort n = 193, validation cohort n = 84) who underwent preoperative MRI were retrospectively analyzed. The risk factors identified through stepwise regression analysis were utilized to construct a clinical model. Radiomics models based on MRI (arterial phase, portal venous phase, delayed phase) across various regions (entire tumor, Peri_5mm, Peri_10mm, Peri_20mm) were developed using the LASSO approach. The features obtained from the intratumoral region and the optimal peritumoral region were combined to design the IntraPeri fusion model. Model performance was assessed using the area under the curve (AUC). Results Larger size, non-smooth margins, and mosaic architecture were risk factors for poorly differentiated HCC (pHCC). The clinical model achieved AUCs of 0.77 and 0.73 in the training and validation cohorts, respectively, while the intratumoral model achieved corresponding AUC values of 0.92 and 0.82. The Peri_10mm model demonstrated superior performance to the Peri_5mm and Peri_20mm models, with AUC values of 0.87 vs. 0.84 vs. 0.73 in the training cohort and 0.80 vs. 0.77 vs. 0.68 in the validation cohort, respectively. The IntraPeri model exhibited remarkable AUC values of 0.95 and 0.86 in predicting pHCC in the training and validation cohorts, respectively. Conclusions Our study highlights the potential of a multiparametric MRI-based radiomic model that integrates intratumoral and peritumoral features as a tool for predicting HCC differentiation. Critical relevance statement Both clinical and multiparametric MRI-based radiomic models, particularly the intratumoral radiomic model, are non-invasive tools for predicting HCC differentiation. Importantly, the IntraPeri fusion model exhibited remarkable predictiveness for individualized HCC differentiation. Key points • Both the intratumoral radiomics model and clinical features were useful for predicting HCC differentiation. • The Peri_10mm radiomics model demonstrated better diagnostic ability than other peritumoral region-based models. • The IntraPeri radiomics fusion model outperformed the other models for predicting HCC differentiation. Graphical Abstract








