Thanks @GBarnesMD for providing a great talk on managing DM in pts w/PAD
#23FellowsAPP @SVM_tweets
🩸 take care of risk factors: ✋🚬; control HTN/DM; add high intensity statin; aspirin 81 mg
🩸add riva 2.5 mg po bid
🩸#flozinate
🩸➡️pcsk9i
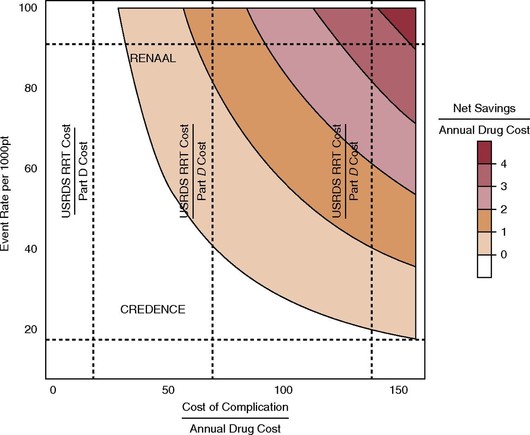
Back then dialysis was more expensive per capita and even under that environment, you had to prior auth to use Cozaar.
The figure compares visually #ARB & #sglt2inhibitors #Flozinate
Payment, Coverage, and Health Economics of SGLT2 Inhibitors
Sodium glucose cotransporter two inhibitors (SGLT2i) slow CKD progression, decrease mortality and heart failure (HF), and have shown evidence for the guided standard of care therapies in patients with or without diabetes (1). Although wide adoption of SGLT2i will provide clinical benefits for patients, whether these agents will ultimately decrease the health care costs of CKD and ESKD is still far from clear. Patients with CKD and type 2 diabetes (T2DM) or HF account for 7% (US$21B) and 4% (US$11.2B) of Medicare fee-for-service expenditures, which far exceeds their representation in the Medicare population, of 5% and 2% respectively (2). Although the cost for treating ESKD has remained stable in inflation-adjusted terms since 2009, it still comprises a significant proportion of Medicare expenditure (about 7%, or US$36B annually) (2). Let us consider the payor’s perspective: “is there robust evidence to justify paying for SGLT2i?” The answer to this requires the examination of four related questions: If these questions are answered affirmatively, then we can ask about the cost of achieving these goals. A comparison with the angiotensin receptor blocker (ARB) trials will also allow us to frame the cost/coverage question using the same pharmacoeconomic arguments (3) that were applied to ARBs when they were still under patent, and thus not widely used. Published trial data about Q1, Q3, and Q4 from Canagliflozin and Renal Events in Diabetes with Established Nephropathy Clinical Evaluation (CREDENCE) (4) and Dapagliflozin and Prevention of Adverse Outcomes in Chronic Kidney Disease (DAPA-CKD) (5) show these drugs outperform ARBs for all outcomes considered (Table 1). QoL …