Delighted to have joined @preLights and to have published my first post! I really enjoyed reading this #preprint on how #ER exit sites regulate the composition of P bodies. I’m excited to discover more about how cells regulate the composition of #biocondensates and how contact sites with them are established and maintained in the future. #ERES #contactsites #preLight #CellBiology #drosophila
#preLight: https://prelights.biologists.com/highlights/the-role-of-er-exit-sites-in-maintaining-p-body-organization-and-transmitting-er-stress-response-during-drosophila-melanogaster-oogenesis/

The role of ER exit sites in maintaining P-body organization and transmitting ER stress response during Drosophila melanogaster oogenesis - preLights
Milano and team shed light on how P bodies are organised at ER exit sites and how ER stress can affect P body composition.
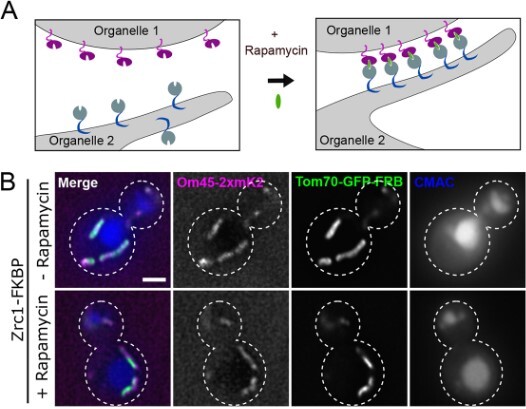

Bringing organelles together mobilises membrane contact tethers
Membrane contacts between organelles are increasingly appreciated as sites of active exchange of molecules that are crucial for intracellular communication. Contact sites are mediated by tethering proteins that bring organelle membranes into close apposition, and a specific inter-organelle contact can often contain multiple tethering proteins. Ayelén González Montoro and colleagues (Amado et al., 2023) here establish inducible contact sites in yeast to study the cross regulation among tethers at specific contact sites. By artificially dimerising organelle-specific transmembrane proteins, they show that formation of mitochondria–vacuole proximity regions is sufficient to recruit and stabilize Vps39, a mobile vacuolar sorting protein known to tether mitochondria–vacuole contacts. The nuclear–vacuolar junction (NVJ) is an endoplasmic reticulum (ER)–vacuole contact site spatially restricted to the perinuclear ER by the tether Nvj1, which interacts with the nuclear membrane. The authors also find that artificially induced contacts between vacuoles and the peripheral ER can recruit other NVJ tether proteins. This indicates that Nvj1 preferentially partitions these tether proteins at the NVJ even when they are able to access the peripheral ER. Taken together, this creative approach reveals the extensive dynamic cross regulation of tether proteins at membrane contacts sites.
