TL;DR: if there is a change in your bowel pattern, at ANY age, ask your doctor if it could be colorectal cancer. Rates of occurrence and death are rising rapidly in the under-65 year age group, some without any risk factors.
#Cancer #ColonCancer #RectalCancer
Colon cancer is on the rise among young people – and research points to one major culprit | Devi Sridhar
#coloncancer #rectalcancer
Colon cancer is on the rise among young people – and research points to one major culprit
If smoking was the cancer villain of the 20th century, eating ultra-processed food may be its 21st-century counterpart, says Prof Devi Sridhar, chair of global public health at the University of Edinburgh
Rectal Cancer Disappears After Experimental Use of Immunotherapy, Now an FDA Designated Breakthrough Therapy
Rectal cancer patients saw their tumors disappear in a clinical trial involving immunotherapy at Memorial Sloan Kettering Cancer Center—without surgery, radiation, or chemotherapy.
Educational Review: Roles of #MRI evaluation of pelvic recurrence in patients with #RectalCancer. (Patricia Perola Dantas et al.)
Roles of MRI evaluation of pelvic recurrence in patients with rectal cancer - Insights into Imaging
Abstract Developments in the multidisciplinary treatment of rectal cancer with advances in preoperative magnetic resonance imaging (MRI), surgical techniques, neoadjuvant chemoradiotherapy, and adjuvant chemotherapy have had a significant impact on patient outcomes, increasing the rates of curative surgeries and reducing pelvic recurrence. Patients with pelvic recurrence have worse prognoses, with an impact on morbidity and mortality. Although local recurrence is more frequent within 2 years of surgical resection of the primary tumor, late recurrence may occur. Clinical manifestations can vary from asymptomatic, nonspecific symptoms, to pelvic pain, bleeding, and fistulas. Synchronous metastatic disease occurs in approximately 50% of patients diagnosed with local recurrence. MRI plays a crucial role in posttreatment follow-up, whether by identifying viable neoplastic tissues or acting as a tool for therapeutic planning and assessing the resectability of these lesions. Locally recurrent tissues usually have a higher signal intensity than muscle on T2-weighted imaging. Thus, attention is required for focal heterogeneous lesions, marked contrast enhancement, early invasive behavior, and asymmetric appearance, which are suspicious for local recurrence. However, postsurgical inflammatory changes related to radiotherapy and fibrosis make it difficult to detect initial lesions. This study therefore aimed to review the main imaging patterns of pelvic recurrence and their implications for the surgical decision-making process. Critical relevance statement MRI plays a crucial role in the posttreatment follow-up of rectal cancer, whether by identifying viable neoplastic tissues or by acting as a tool for therapeutic planning. This study reviewed the main imaging patterns of pelvic recurrence. Key Points MRI aids in surgical planning and the detection of pelvic recurrence and postoperative complications. Being familiar with surgical techniques enables radiologists to identify expected MRI findings. Patterns of rectal cancer recurrence have been categorized by pelvic compartments. Neoplastic tissue may mimic postsurgical and postradiotherapy changes. Resectability of pelvic recurrence is highly related to lesion location. Graphical Abstract
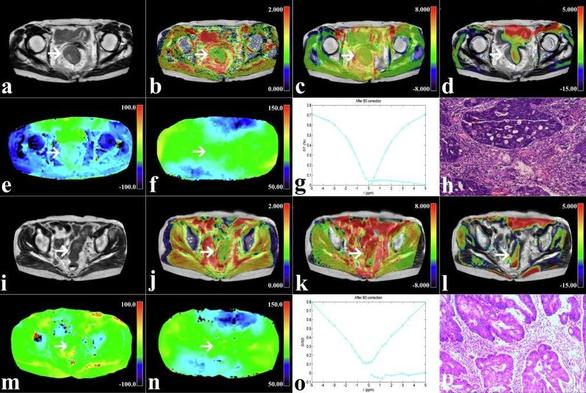
Nan Meng et al. show that glucose chemical exchange saturation transfer (glucoCEST) MRI, in combination with amide proton transfer-weighted imaging (APTWI) and diffusion-weighted imaging (DWI), can effectively differentiate between high- and low-grade #RectalCancer, which could be a promising imaging marker.
Glucose chemical exchange saturation transfer MRI for predicting the histological grade of rectal cancer: a comparative study with amide proton transfer-weighted and diffusion-weighted imaging - Insights into Imaging
Background To evaluate the utility of glucose chemical exchange saturation transfer (glucoCEST) MRI with non-contrast injection in predicting the histological grade of rectal cancer. Methods This prospective analysis included 60 patients with preoperative rectal cancer who underwent pelvic glucoCEST, amide proton transfer-weighted imaging (APTWI), and diffusion-weighted imaging (DWI). In total, 21 low-grade and 39 high-grade cases were confirmed by postoperative pathology. The MTRasym (1.2 ppm), MTRasym (3.5 ppm), and apparent diffusion coefficient (ADC) values of lesions between the low-grade and high-grade groups were compared. The area under the receiver operating characteristic curve (AUC) was generated to evaluate the diagnostic performance of each technique. Logistic regression (LR) analysis was applied to determine independent predictors and for multi-parameter combined diagnosis. Results Elevated MTRasym (1.2 ppm), MTRasym (3.5 ppm) values and lower ADC values were observed in the high-grade group compared with low-grade cases (all p < 0.01). The AUCs of MTRasym (1.2 ppm), MTRasym (3.5 ppm), and ADC for differentiating between low- and high-grade rectal cancer cases were 0.792, 0.839, and 0.855, respectively. The diagnostic performance of the combination of the three indexes was improved (AUC, 0.969; sensitivity, 95.24%; specificity, 87.18%). The good consistency and reliability of the combination of independent predictors were demonstrated by calibration curve analysis and DCA. Conclusion The glucoCEST MRI without contrast injection, APTWI, and DWI all facilitate the assessment of histological grade in rectal cancer, and the combination of the three can effectively discriminate between high- and low-grade rectal cancer, which is expected to be a promising imaging marker. Critical relevance statement The glucose chemical exchange saturation transfer MRI method facilitates the assessment of histological grade in rectal cancer and offers additional information to improve the diagnostic performance of amide proton transfer-weighted imaging, and diffusion-weighted imaging. Key Points Glucose chemical exchange saturation transfer imaging could differentiate histological grade. Amide proton transfer-weighted and diffusion-weighted were associated with histological grade. The combination of different parameters showed the best diagnostic performance. Graphical Abstract
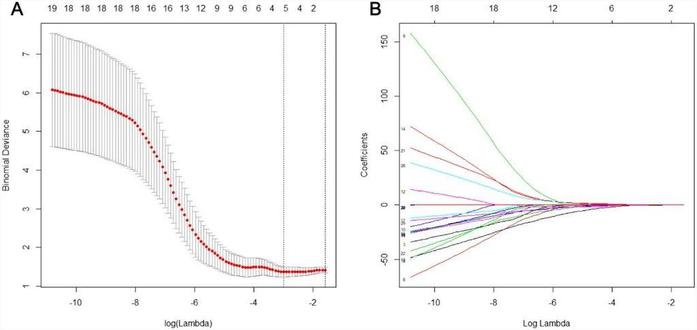
Junjie Fang et al. developed this assessment model by texture analysis for the preoperative prediction of extramural vascular invasion (EMVI), determining that the model showed excellent performance.
Value of texture analysis based on dynamic contrast-enhanced magnetic resonance imaging in preoperative assessment of extramural venous invasion in rectal cancer - Insights into Imaging
Objective Accurate preoperative assessment of extramural vascular invasion (EMVI) is critical for the treatment and prognosis of rectal cancer. The aim of our research was to develop an assessment model by texture analysis for preoperative prediction of EMVI. Materials and methods This study enrolled 44 rectal patients as train cohort, 7 patients as validation cohort and 18 patients as test cohort. A total of 236 texture features from DCE MR imaging quantitative parameters were extracted for each patient (59 features of Ktrans, Kep, Ve and Vp), and key features were selected by least absolute shrinkage and selection operator regression (LASSO). Finally, clinical independent risk factors, conventional MRI assessment, and T-score were incorporated to construct an assessment model using multivariable logistic regression. Results The T-score calculated using the 4 selected key features were significantly correlated with EMVI (p < 0.010). The area under the receiver operating characteristic curve (AUC) was 0.797 for discriminating between EMVI-positive and EMVI-negative patients with a sensitivity of 88.2% and specificity of 70.4%. The conventional MRI assessment of EMVI had a sensitivity of 23.53% and a specificity of 96.30%. The assessment model showed a greatly improved performance with an AUC of 0.954 (sensitivity, 88.2%; specificity, 92.6%) in train cohort, 0.833 (sensitivity, 66.7%; specificity, 100%) in validation cohort and 0.877 in test cohort, respectively. Conclusions The assessment model showed an excellent performance in preoperative assessment of EMVI. It demonstrates strong potential for improving the accuracy of EMVI assessment and provide a reliable basis for individualized treatment decisions.
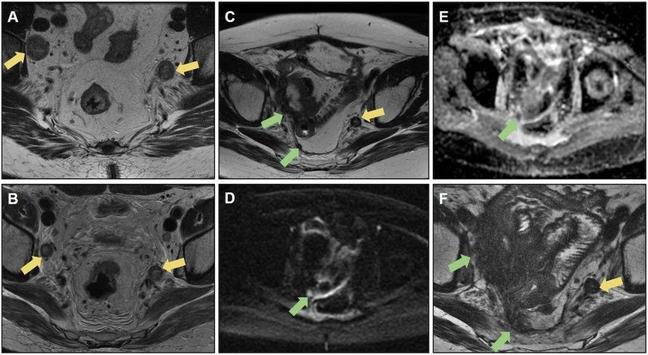
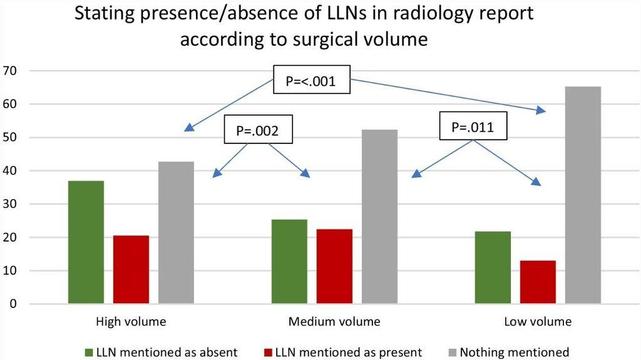
Retrospective study assessed whether lateral lymph nodes (LLNs) were mentioned in primary #MRI reports, discovering that in almost half of of the reports for patients with advanced #RectalCancer, LLNs were not mentioned. (Tania C. Sluckin et al.)
Retrospective evaluation of national MRI reporting quality for lateral lymph nodes in rectal cancer patients and concordance with prospective re-evaluation following additional training - Insights into Imaging
Objectives The presence and size of lateral lymph nodes (LLNs) are important factors influencing treatment decisions for rectal cancer. Awareness of the clinical relevance and describing LLNs in MRI reports is therefore essential. This study assessed whether LLNs were mentioned in primary MRI reports at a national level and investigated the concordance with standardised re-review. Methods This national, retrospective, cross-sectional cohort study included 1096 patients from 60 hospitals treated in 2016 for primary cT3-4 rectal cancer ≤ 8 cm from the anorectal junction. Abdominal radiologists re-reviewed all MR images following a 2-h training regarding LLNs. Results Re-review of MR images identified that 41.0% of enlarged (≥ 7 mm) LLNs were not mentioned in primary MRI reports. A contradictory anatomical location was stated for 73.2% of all LLNs and a different size (≥/< 7 mm) for 41.7%. In total, 49.4% of all cases did not mention LLNs in primary MRI reports. Reporting LLNs was associated with stage (cT3N0 44.3%, T3N+/T4 52.8%, p = 0.013), cN stage (N0 44.1%, N1 48.6%, N2 59.5%, p < 0.001), hospital type (non-teaching 34.6%, teaching 52.2%, academic 53.2% p = 0.006) and annual rectal cancer resection volumes (low 34.8%, medium 47.7%, high 57.3% p < 0.001). For LLNs present according to original MRI reports (n = 226), 64.2% also mentioned a short-axis size, 52.7% an anatomical location and 25.2% whether it was deemed suspicious. Conclusions Almost half of the primary MRI reports for rectal cancer patients treated in the Netherlands in 2016 did not mention LLNs. A significant portion of enlarged LLNs identified during re-review were also not mentioned originally, with considerable discrepancies for location and size. These results imply insufficient awareness and indicate the need for templates, education and training.
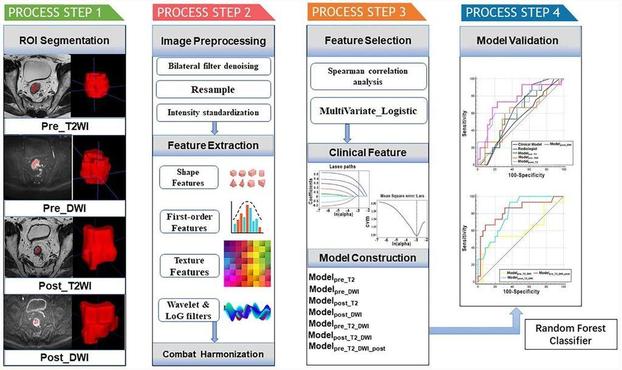
A new study by Qiurong Wei et al. looks at the construction and validation of multiparametric MR-based #radiomics models to predict lymph node metastasis following neoadjuvant chemoradiotherapy (nCRT) in patients with locally advanced #RectalCancer (LARC).
Multiparametric MRI-based radiomic model for predicting lymph node metastasis after neoadjuvant chemoradiotherapy in locally advanced rectal cancer - Insights into Imaging
Objectives To construct and validate multiparametric MR-based radiomic models based on primary tumors for predicting lymph node metastasis (LNM) following neoadjuvant chemoradiotherapy (nCRT) in locally advanced rectal cancer (LARC) patients. Methods A total of 150 LARC patients from two independent centers were enrolled. The training cohort comprised 100 patients from center A. Fifty patients from center B were included in the external validation cohort. Radiomic features were extracted from the manually segmented volume of interests of the primary tumor before and after nCRT. Feature selection was performed using multivariate logistic regression analysis. The clinical risk factors were selected via the least absolute shrinkage and selection operator method. The radiologist’s assessment of LNM was performed. Eight models were constructed using random forest classifiers, including four single-sequence models, three combined-sequence models, and a clinical model. The models’ discriminative performance was assessed via receiver operating characteristic curve analysis quantified by the area under the curve (AUC). Results The AUCs of the radiologist’s assessment, the clinical model, and the single-sequence models ranged from 0.556 to 0.756 in the external validation cohort. Among the single-sequence models, modelpost_DWI exhibited superior predictive power, with an AUC of 0.756 in the external validation set. In combined-sequence models, modelpre_T2_DWI_post had the best diagnostic performance in predicting LNM after nCRT, with a significantly higher AUC (0.831) than those of the clinical model, modelpre_T2_DWI, and the single-sequence models (all p < 0.05). Conclusions A multiparametric model that incorporates MR radiomic features before and after nCRT is optimal for predicting LNM after nCRT in LARC. Critical relevance statement This study enrolled 150 LARC patients from two independent centers and constructed multiparametric MR-based radiomic models based on primary tumors for predicting LNM following nCRT, which aims to guide therapeutic decisions and predict prognosis for LARC patients. Key Points The biological characteristics of primary tumors and metastatic LNs are similar in rectal cancer. Radiomics features and clinical data before and after nCRT provide complementary tumor information. Preoperative prediction of LN status after nCRT contributes to clinical decision-making. Graphical Abstract
🎉 100% Cancer Remission:
A groundbreaking study has achieved a 100% cancer remission rate in all rectal cancer patients through a monoclonal antibody trial, as reported by Memorial Sloan Kettering (MSK) Cancer Center.
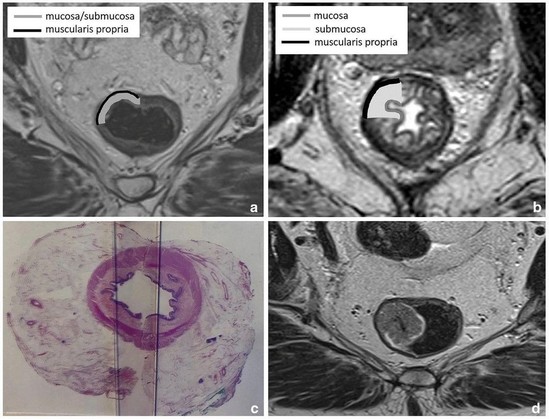
🥈The 2nd Most Downloaded article of 2023 is by Nino Bogveradze et al. with over 20k downloads! This article provides an image-based overview of key anatomical concepts for treatment planning, response evaluation, and post-operative assessment related to #RectalCancer.
Congratulations!
MRI anatomy of the rectum: key concepts important for rectal cancer staging and treatment planning - Insights into Imaging
A good understanding of the MRI anatomy of the rectum and its surroundings is pivotal to ensure high-quality diagnostic evaluation and reporting of rectal cancer. With this pictorial review, we aim to provide an image-based overview of key anatomical concepts essential for treatment planning, response evaluation and post-operative assessment. These concepts include the cross-sectional anatomy of the rectal wall in relation to T-staging; differences in staging and treatment between anal and rectal cancer; landmarks used to define the upper and lower boundaries of the rectum; the anatomy of the pelvic floor and anal canal, the mesorectal fascia, peritoneum and peritoneal reflection; and guides to help discern different pelvic lymph node stations on MRI to properly stage regional and non-regional rectal lymph node metastases. Finally, this review will highlight key aspects of post-treatment anatomy, including the assessment of radiation-induced changes and the evaluation of the post-operative pelvis after different surgical resection and reconstruction techniques.