✨ Pilot study highlight:
Using advanced CUDI processing (regularization, motion correction, SVD filtering), we showed that spatiotemporal similarity analysis outperforms TIC fitting in distinguishing RCC tumors from normal parenchyma.
A promising step toward more accurate, noninvasive RCC diagnosis.#Ultrasound
#CUDI #Radiomics #RCC #KidneyCancer #MedicalImaging #ContrastEnhancedUltrasound #AIinMedicine #DeepLearning #OncoImaging #ImagingInnovation #MedTech #ResearchUpdate⚛️ How do graph-driven radiomic networks improve our ability to stratify cancer patients?
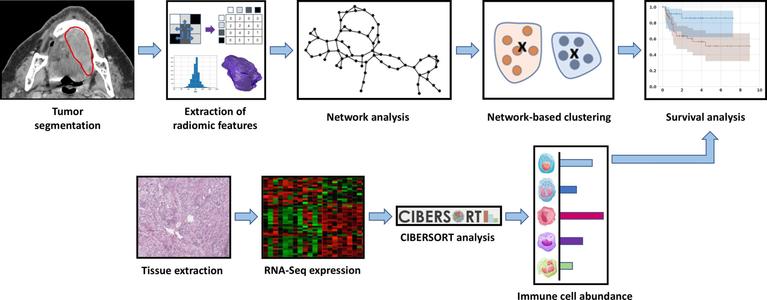
🔗 Radiomic clustering using graph network techniques coupled with unbalanced optimal transport. Computational and Structural Biotechnology Journal, DOI: https://doi.org/10.1016/j.csbj.2025.10.066
📚 CSBJ: https://www.csbj.org/
#Radiomics #MedicalImaging #CancerResearch #PrecisionOncology #AIinHealthcare #MachineLearning #Radiogenomics #DigitalHealth #ComputationalOncology #OncologyResearch #Bioinformatics #MedicalAI
Triple-negative breast cancer is tough to predict due to its complexity. New research published in
#SPIE_JMI shows that even unstable radiomic features from MRI scans can still be highly predictive.
Read more in the
#OpenAccess article:
https://bit.ly/47aiW6U
#MedicalImaging #Radiomics
Neue Forschungskooperation: Die Zukunft der biomedizinischen Bildanalyse
Development of an Artificial Intelligence System for Distinguishing Malignant from Benign Soft-Tissue Tumors Using Contrast-Enhanced MR Images
<p xmlns:xsi="http://www.w3.org/2001/XMLSchema-instance" dir="auto" id="d2621084e267">
<b>
<i>Introduction:</i>
</b> The integration of artificial intelligence (AI) into orthopedics has enhanced the
diagnosis of various conditions; however, its use in diagnosing soft-tissue tumors
remains limited owing to its complexity. This study aimed to develop and assess an
AI-driven diagnostic support system for magnetic resonance imaging (MRI)-based soft-tissue
tumor diagnosis, potentially improving accuracy and aiding radiologists and orthopedic
surgeons.
<b>
<i>Methods:</i>
</b> An experienced orthopedic oncologist and radiologist annotated 720 images from 77
cases (41 benign and 36 malignant soft-tissue tumors). Eleven tumor subtypes were
identified and classified into benign and malignant groups based on histological diagnosis.
Utilizing the standard machine learning classifier pipeline, we examined and down-selected
imaging protocols and their predominant radiomic features within the tumor’s three-dimensional
region to differentiate between benign and malignant tumors. Among the scan protocols,
contrast-enhanced T1-weighted fat-suppressed images showed the most accurate classification
based on radiomic features. We focused on the two-dimensional features from the largest
tumor boundary surface and its neighboring slices, leveraging texture-based radiomic
and deep convolutional neural network features from a pretrained VGG19 model.
<b>
<i>Results:</i>
</b> The test data comprised 44 contrast-enhanced images (22 benign and 22 malignant soft-tissue
tumors) containing six malignant and five benign subtypes distinct from the training
data. We compared expert and nonexpert human performances against AI by assessing
malignancy detection and the time required for classification. The AI model showed
comparable accuracy (AUC 0.91) to that of radiologists (AUC 0.83) and orthopedic surgeons
(AUC 0.73). Notably, the AI model processed data approximately 400 times faster than
its human counterparts, showcasing its capacity to significantly boost diagnostic
efficiency.
<b>
<i>Conclusion:</i>
</b> We developed an AI-driven diagnostic support system for MRI-based soft-tissue tumor
diagnosis. While additional refinement is necessary for clinical applications, our
system has exhibited promising potential in differentiating between benign and malignant
soft-tissue tumors based on MRI.
</p><p xmlns:xsi="http://www.w3.org/2001/XMLSchema-instance" dir="auto" id="d2621084e296">MRI is invaluable for the qualitative diagnosis of soft-tissue tumors, but radiologists
face challenges despite advancements in imaging technologies. This study aimed to
develop and evaluate an AI-driven diagnostic support system for MRI-based soft-tissue
tumor diagnosis. The study used 720 images from 77 cases. Using a standard machine
learning classifier pipeline, we examined and down-selected imaging protocols and
their predominant radiomic features within each tumor’s three-dimensional region to
differentiate between benign and malignant tumors. Contrast-enhanced and T2-weighted
images were identified as candidates for developing a machine learning model for malignancy
classification. We focused on the two-dimensional features from the largest tumor
boundary surface and its neighboring slices, leveraging texture-based radiomic and
deep convolutional neural network features from a pretrained VGG19 model. A support
vector machine classifier trained with contrast-enhanced images demonstrated the best
performance in preliminary validation investigations. Validation was performed using
test data comprising 44 contrast-enhanced images. The AI model exhibited comparable
accuracy (AUC 0.91) to that of radiologists (AUC 0.83) and orthopedic surgeons (AUC
0.73). The AI model processed data approximately 400 times faster than its human counterparts,
demonstrating its capacity to significantly enhance diagnostic efficiency. While additional
refinement is necessary for clinical applications, our system has exhibited promising
potential in differentiating between benign and malignant soft-tissue tumors based
on MR images.
</p>
#Radiomics, an emerging machine learning approach for conventional
#MedicalImaging, is applied to conjunctiva photos to analyze morphological alterations in the microvasculature beyond direct visualization in school-aged children.
Learn more:
www.spiedigitallibra...
#BiophotonicsDiscovery A CT #radiomics-based ML model developed by Zongjie Wei et al. can noninvasively predict HER2 status in #BladderCancer with high accuracy and interpretability, aiding in the preoperative decision-making process.
#InsightsIntoImaging
🔗 https://buff.ly/3AEBAaO


A radiomics-based interpretable machine learning model to predict the HER2 status in bladder cancer: a multicenter study - Insights into Imaging
Objective To develop a computed tomography (CT) radiomics-based interpretable machine learning (ML) model to preoperatively predict human epidermal growth factor receptor 2 (HER2) status in bladder cancer (BCa) with multicenter validation. Methods In this retrospective study, 207 patients with pathologically confirmed BCa were enrolled and divided into the training set (n = 154) and test set (n = 53). Least absolute shrinkage and selection operator (LASSO) regression was used to identify the most discriminative features in the training set. Five radiomics-based ML models, namely logistic regression (LR), support vector machine (SVM), k-nearest neighbors (KNN), eXtreme Gradient Boosting (XGBoost) and random forest (RF), were developed. The predictive performance of established ML models was evaluated by the area under the receiver operating characteristic curve (AUC). The Shapley additive explanation (SHAP) was used to analyze the interpretability of ML models. Results A total of 1218 radiomics features were extracted from the nephrographic phase CT images, and 11 features were filtered for constructing ML models. In the test set, the AUCs of LR, SVM, KNN, XGBoost, and RF were 0.803, 0.709, 0.679, 0.794, and 0.815, with corresponding accuracies of 71.7%, 69.8%, 60.4%, 75.5%, and 75.5%, respectively. RF was identified as the optimal classifier. SHAP analysis showed that texture features (gray level size zone matrix and gray level co-occurrence matrix) were significant predictors of HER2 status. Conclusions The radiomics-based interpretable ML model provides a noninvasive tool to predict the HER2 status of BCa with satisfactory discriminatory performance. Critical relevance statement An interpretable radiomics-based machine learning model can preoperatively predict HER2 status in bladder cancer, potentially aiding in the clinical decision-making process. Key Points The CT radiomics model could identify HER2 status in bladder cancer. The random forest model showed a more robust and accurate performance. The model demonstrated favorable interpretability through SHAP method. Graphical Abstract
An interpretable #DeepLearning #radiomics (IDLR) model based on [18F]FDG PET images accurately diagnosed #AlzheimersDisease (AD) and predicted progression from mild cognitive impairment (MCI) to AD, with over 76% accuracy. Jiehui Jiang et al. found it outperformed traditional models and identified key features linked to cognitive decline.
#EuropeanRadiology
🔗 https://buff.ly/4hM533e
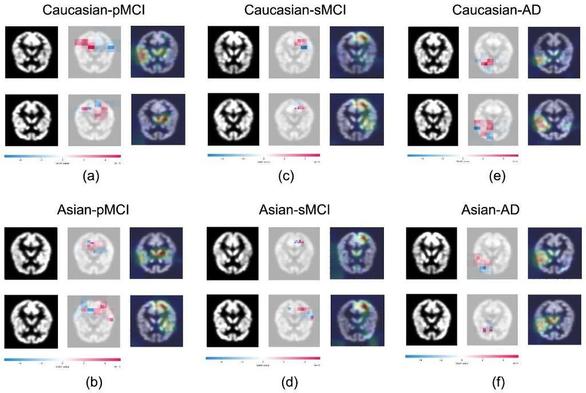

Using interpretable deep learning radiomics model to diagnose and predict progression of early AD disease spectrum: a preliminary [18F]FDG PET study - European Radiology
Objectives In this study, we propose an interpretable deep learning radiomics (IDLR) model based on [18F]FDG PET images to diagnose the clinical spectrum of Alzheimer’s disease (AD) and predict the progression from mild cognitive impairment (MCI) to AD. Methods This multicentre study included 1962 subjects from two ethnically diverse, independent cohorts (a Caucasian cohort from ADNI and an Asian cohort merged from two hospitals in China). The IDLR model involved feature extraction, feature selection, and classification/prediction. We evaluated the IDLR model’s ability to distinguish between subjects with different cognitive statuses and MCI trajectories (sMCI and pMCI) and compared results with radiomic and deep learning (DL) models. A Cox model tested the IDLR signature’s predictive capability for MCI to AD progression. Correlation analyses identified critical IDLR features and verified their clinical diagnostic value. Results The IDLR model achieved the best classification results for subjects with different cognitive statuses as well as in those with MCI with distinct trajectories, with an accuracy of 76.51% [72.88%, 79.60%], (95% confidence interval, CI) while those of radiomic and DL models were 69.13% [66.28%, 73.12%] and 73.89% [68.99%, 77.89%], respectively. According to the Cox model, the hazard ratio (HR) of the IDLR model was 1.465 (95% CI: 1.236–1.737, p < 0.001). Moreover, three crucial IDLR features were significantly different across cognitive stages and were significantly correlated with cognitive scale scores (p < 0.01). Conclusions Preliminary results demonstrated that the IDLR model based on [18F]FDG PET images enhanced accuracy in diagnosing the clinical spectrum of AD. Key Points Question The study addresses the lack of interpretability in existing DL classification models for diagnosing the AD spectrum. Findings The proposed interpretable DL radiomics model, using radiomics-supervised DL features, enhances interpretability from traditional DL models and improves classification accuracy. Clinical relevance The IDLR model interprets DL features through radiomics supervision, potentially advancing the application of DL in clinical classification tasks.
Critical Review: The prognostic value of radiogenomics using CT in patients with #LungCancer. (Yixiao Jiang et al.)
#InsightsIntoImaging #Radiomics
🔗 https://buff.ly/4hFte36
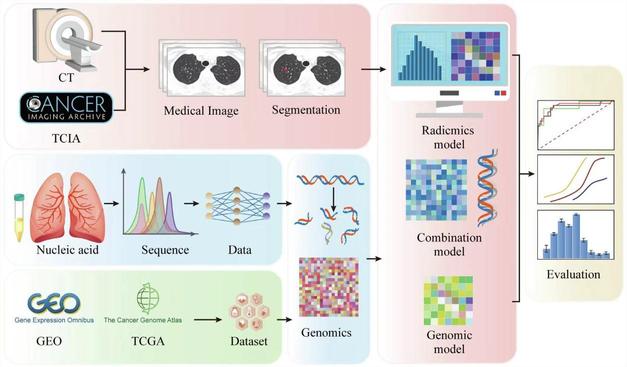

The prognostic value of radiogenomics using CT in patients with lung cancer: a systematic review - Insights into Imaging
Abstract This systematic review aimed to evaluate the effectiveness of combining radiomic and genomic models in predicting the long-term prognosis of patients with lung cancer and to contribute to the further exploration of radiomics. This study retrieved comprehensive literature from multiple databases, including radiomics and genomics, to study the prognosis of lung cancer. The model construction consisted of the radiomic and genomic methods. A comprehensive bias assessment was conducted, including risk assessment and model performance indicators. Ten studies between 2016 and 2023 were analyzed. Studies were mostly retrospective. Patient cohorts varied in size and characteristics, with the number of patients ranging from 79 to 315. The construction of the model involves various radiomic and genotic datasets, and most models show promising prediction performance with the area under the receiver operating characteristic curve (AUC) values ranging from 0.64 to 0.94 and the concordance index (C-index) values from 0.28 to 0.80. The combination model typically outperforms the single method model, indicating higher prediction accuracy and the highest AUC was 0.99. Combining radiomics and genomics in the prognostic model of lung cancer may improve the predictive performance. However, further research on standardized data and larger cohorts is needed to validate and integrate these findings into clinical practice. Critical relevance statement The combination of radiomics and genomics in the prognostic model of lung cancer improved prediction accuracy in most included studies. Key Points The combination of radiomics and genomics can improve model performance in most studies. The results of establishing prognosis models by different methods are discussed. The combination of radiomics and genomics may be helpful to provide better treatment for patients. Graphical Abstract
An all-new addition to the #ESREssentials series dives into the practice recommendations by EuSoMII regarding the promising tool of #radiomics. How can you as a radiologist best utilize this technology? (João Santinha et al.)
#EuropeanRadiology
🔗 https://buff.ly/3UG52E4


ESR Essentials: radiomics—practice recommendations by the European Society of Medical Imaging Informatics - European Radiology
Abstract Radiomics is a method to extract detailed information from diagnostic images that cannot be perceived by the naked eye. Although radiomics research carries great potential to improve clinical decision-making, its inherent methodological complexities make it difficult to comprehend every step of the analysis, often causing reproducibility and generalizability issues that hinder clinical adoption. Critical steps in the radiomics analysis and model development pipeline—such as image, application of image filters, and selection of feature extraction parameters—can greatly affect the values of radiomic features. Moreover, common errors in data partitioning, model comparison, fine-tuning, assessment, and calibration can reduce reproducibility and impede clinical translation. Clinical adoption of radiomics also requires a deep understanding of model explainability and the development of intuitive interpretations of radiomic features. To address these challenges, it is essential for radiomics model developers and clinicians to be well-versed in current best practices. Proper knowledge and application of these practices is crucial for accurate radiomics feature extraction, robust model development, and thorough assessment, ultimately increasing reproducibility, generalizability, and the likelihood of successful clinical translation. In this article, we have provided researchers with our recommendations along with practical examples to facilitate good research practices in radiomics. Key Points Radiomics’ inherent methodological complexity should be understood to ensure rigorous radiomic model development to improve clinical decision-making. Adherence to radiomics-specific checklists and quality assessment tools ensures methodological rigor. Use of standardized radiomics tools and best practices enhances clinical translation of radiomics models.