https://www.biorxiv.org/content/10.64898/2026.05.15.725329v1?rss=1 #Elasticity #Mechanical #Cell
https://www.biorxiv.org/content/10.64898/2026.05.15.725329v1?rss=1 #Elasticity #Mechanical #Cell
https://doi.org/doi:10.1103/qtcv-gk3y
https://pubmed.ncbi.nlm.nih.gov/42141626/
#Elasticity #Cell
https://www.biorxiv.org/content/10.64898/2026.04.09.717535v1?rss=1 #Elasticity #Cell
https://doi.org/doi:10.1371/journal.pcbi.1013532
https://pubmed.ncbi.nlm.nih.gov/41926455/
#Elasticity #Pressure #Cell
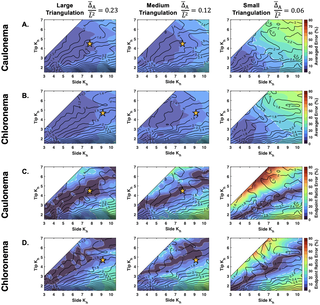
A surface morphology-based inference method for the cell wall elasticity profile in tip-growing cells
Author summary Tip-growing cells can be characterized by their fast growth concentrated at the cell’s apex. Their growth and morphogenesis are tightly regulated processes involving cell wall addition and rearrangement while the cell wall is under stress originating from the cell’s internal turgor pressure. We start by studying the cell wall’s elastic properties, one aspect of the cell growth process. We use a method of marker point tracking across the surface of the tip-growing cell to measure the wall’s elasticity profile. In this work, we present a parameter sensitivity study of this method on synthetic cells and report our results on experimental moss tip-growing cells. Our results suggest that this inference method can reliably measure a cell wall elasticity gradient under combined geometric and mechanical conditions that create elastic strains within 5% at the tip.
https://doi.org/doi:10.3390/membranes16030095
https://pubmed.ncbi.nlm.nih.gov/41893281/
#Elasticity #Cell
- some charged attacks I dreamt he had, aerial, heavy, and light! Fighting the Hip Chip Clan and the Silent Roar gangs in this one
#spaceRARt #scalieart #human #superhero #elasticity #gamelogic
https://arxiv.org/abs/2603.19723 #Cond-Mat.Soft #Extracellular #Elasticity #Q-Bio.To

Modelling the passive and active response of skeletal muscles within the adapted Voigt representation framework
We present a constitutive model for the passive and active response of skeletal muscles. At variance with more classical approaches, the model is developed exploiting adapted Voigt representations of strain and stress tensors within the context of nonlinear Cauchy elasticity. This framework allows us to identify non-trivial stress-strain relations in a rather direct way from experimental data, enhancing the mechanical interpretability of the material functions that describe the tissue response and obtaining additional insight on the distinct role of the contractile fibres and of the surrounding extracellular matrix. We propose a two-material model, with an additive splitting of the stress contributions, in which only one component depends on an activation parameter. The constitutive model for the passive behaviour satisfactorily predicts the nonlinear stress response to elongation at different relative orientations with respect to the fibre direction and highlights the dominant role of the extracellular matrix. The activation model, essentially determined by the mechanics of the contractile fibres, captures well the isometric stress response through the prescription of an elasto-plastic evolution of the along-fibre active strain.
https://www.biorxiv.org/content/10.64898/2026.03.14.711786v1?rss=1 #Elasticity #Lamin

Inter–lamin interactions control meshwork topology in a polymer–gel model of nuclear lamina
The nuclear lamina, composed of supramolecular structures of lamin proteins, is a two–dimensional protein meshwork that preserves the structural integrity, elasticity, and morphology of the nucleus. Lamins—A/C–type and B–type—assemble into dynamic, individual but interacting networks with distinct structural properties. Lamina meshwork assembly can be disrupted by lamin mutations in diseases known as laminopathies. Despite extensive experimental insights, the biophysical mechanisms that alter the lamina meshwork topology in health and disease remain relatively poorly understood. In this study, we develop a coarse-grained molecular dynamics (MD) model of lamina self–assembly, where lamin dimers are modeled as semiflexible polymers confined within an elastic nuclear shell. By systematically interrogating inter–lamin and lamin–shell association affinities, our simulations reproduce a plethora of experimentally observed lamina architectures, from lattice–like to fibrous meshwork topologies. This elucidates how the interplay between inter–lamin and lamin–nuclear envelope interactions can shape the nuclear lamina. Importantly, inter–lamin interactions can cause a heterogeneous distribution of lamins on the surface and result in large, lamin–free surface domains at sufficiently low lamin-shell affinities. Furthermore, paracrystalline lamin sheets form with increasing propensity for parallel lamin alignment, in addition to the canonical, sticky terminal groups. Overall, our integrative MD and network analysis provide the first explicit polymer physics model of the lamina and demonstrate how lamin interactions may affect the mesoscale architecture of the lamina in disease. ### Competing Interest Statement The authors have declared no competing interest. Scientific and Technological Research Council of Turkey, https://ror.org/04w9kkr77, 124N935
https://arxiv.org/abs/2603.05533 #Cond-Mat.Mtrl-Sci #Physics.Class-Ph #Elasticity #Matrix

Universal Displacements in Linear Strain-Gradient Elasticity
We study universal displacement fields in three-dimensional linear strain-gradient elasticity within the Toupin-Mindlin first strain-gradient theory. Building on the approach of Yavari (2020), we derive, for each material symmetry class, the universality PDEs obtained by requiring the equilibrium equations (in the absence of body forces) to hold for any material in that class, and we determine the complete set of universal displacements. Using the full symmetry classification together with compact matrix representations of the elasticity tensors, we provide explicit characterizations for all 48 strain-gradient symmetry classes, including centrosymmetric and chiral classes. For several high-symmetry classes, the strain-gradient universality PDEs impose no additional restrictions beyond the classical ones, so the universal displacement families coincide with those of classical linear elasticity (for example, the isotropic classes SO(3) and O(3)). For lower symmetry classes, the strain-gradient universality PDEs can be stricter than their classical counterparts, so the universal displacements form proper subsets of the classical universal displacement families due to additional higher-order differential conditions.
https://doi.org/doi:10.1111/omi.70025
https://pubmed.ncbi.nlm.nih.gov/41782230/
#Elasticity #Cell
