📰 "Stiff-FCS: Single-Cell Stiffness Profiling With Integrated Molecular and Functional Analysis"
https://arxiv.org/abs/2604.00467 #Q-Bio.Cb
#Lamin
Stiff-FCS: Single-Cell Stiffness Profiling With Integrated Molecular and Functional Analysis
Cell stiffness is a key determinant of how cells deform, migrate, and adapt to mechanically restrictive environments, yet existing single-cell stiffness assays remain difficult to combine with molecular analysis and downstream functional studies. To address these limitations, we introduce a microfluidic platform, stiffness-based ferrohydrodynamic cell sorting (Stiff-FCS), designed for high-throughput quantification of single-cell stiffness, on-chip molecular analysis, and post-assay cell recovery. Stiff-FCS combines ferrofluid-driven actuation with graded confinement channels to control cell movement, induce deformation, and spatially separate cells based on stiffness. An inverse computational model converts cell position and morphology into quantitative Young's modulus values. We demonstrate stiffness profiling of hundreds to thousands of cells per chip within minutes, same-cell fluorescence-based protein analysis, and recovery of stiffness-defined cells for downstream assays. Across diverse human and mouse cell lines, Lamin A/C showed the most consistent association with stiffness, whereas softer cells exhibited greater migratory capacity than stiffer cells. In a series of human head and neck cancer cell models, Stiff-FCS further resolved a stiff, less migratory subpopulation enriched in a higher-molecular-weight Vimentin state, offering a workflow for linking single-cell stiffness to molecular heterogeneity and cell behavior.
📰 "Matrix stiffness induces midnolin-dependent lamin B1 degradation to control myoblast differentiation"
https://doi.org/doi:10.1038/s44319-026-00753-0https://pubmed.ncbi.nlm.nih.gov/41917260/ #Lamin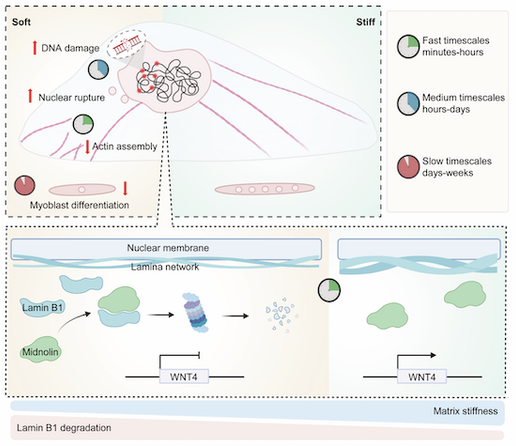
Matrix stiffness induces midnolin-dependent lamin B1 degradation to control myoblast differentiation - EMBO Reports
Cells decode mechanical cues to direct fate decisions through nuclear remodeling, yet nuclear adaptors to mechanical signals remain elusive. Here, we show that soft matrix suppresses myoblast differentiation and induces nuclear abnormality within 30 min, accompanied by a greater than 60% reduction in lamin B1 proteins levels. Mechanistically, midnolin interacts with lamin B1 and mediates ubiquitination-independent degradation of lamin B1 on soft matrix, through the Catch domain of midnolin engaging a β-strand within lamin B1’s Ig-like domain. Functionally, moderate lamin B1 expression is essential for myoblast differentiation initiation, as its depletion either by siRNA or CRISPR knockout abolishes myogenic capacity. Our findings reveal that the midnolin-proteasome axis directly converts mechanical inputs into lineage commitment by triggering lamin B1 degradation, defining a novel nuclear mechano-adaptation pathway.
📰 "B-type lamins maintain transcriptional homeostasis by spatially controlling chromatin-speckle proximity"
https://doi.org/doi:10.1093/nar/gkag283https://pubmed.ncbi.nlm.nih.gov/41909953/ #Lamin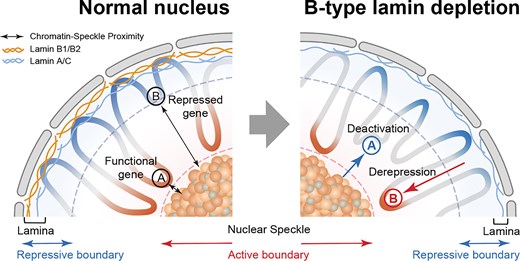
B-type lamins maintain transcriptional homeostasis by spatially controlling chromatin–speckle proximity
Abstract. B-type lamins are essential nuclear scaffold proteins known to maintain peripheral heterochromatin through lamina-associated domains (LADs). Here
📰 "STING causes replication stress and nascent DNA degradation via SAMHD1"
https://www.biorxiv.org/content/10.64898/2026.03.28.714577v1?rss=1 #Lamin
STING causes replication stress and nascent DNA degradation via SAMHD1
STING is a key innate immune adaptor, classically activated by cytosolic DNA via cGAS-cGAMP to induce type I interferon signaling. While its cytoplasmic role is well-defined, recent studies reveal that STING participates in non-canonical signaling pathways and localizes at the nuclear envelope and chromatin, where its functions remain poorly understood. In Hutchinson Gilford Progeria Syndrome (HGPS), a premature aging caused by expression of lamin A mutant protein named progerin, STING accumulates in the nucleus and drives chronic inflammation. Here, we report that replication stress (RS) is a trigger of STING nuclear accumulation and binding to chromatin. In addition, we uncover a previously unrecognized role for nuclear STING binding to nascent DNA and promoting RS in progeria and tumor cells. Mechanistically, STING contributes to replication fork slowing and stalling by limiting dNTPs availabilithy. In addition, STING hinders replication fork protection/stability upon stalling, by facilitating MRE11-mediated nascent DNA degradation (NDD). We also find taht STING contribution to depletion of dNTPs and NDD is mediated by SAMHD1. As such, SAMHD1 knockdown phenocopies STING abrogation in progeria cells and rescues replication fork speed and stability in STING-overexpressing tumor cells. These findings define a pathological STING-SAMHD1 axis that drives RS and genome instability in both progeria cells and tumor cells with elevated STING activity, uncovering a feedforward loop between innate immune signaling and impaired replication.
### Competing Interest Statement
The authors have declared no competing interest.
National Institutes of Health, https://ror.org/01cwqze88, RAG082759A, RAG076145A, R01AG058714
Glenn Foundation for Medical Research Postdoctoral Fellowship in Aging Research, PD24164
📰 "Lamin A/C as a Molecular Link Between Nuclear Organization, Chromatin Dynamics, and Tumor Progression"
https://doi.org/doi:10.3390/cells15060501https://pubmed.ncbi.nlm.nih.gov/41892292/ #Mechanics #Dynamics #Lamin
The cytoskeleton contributes to abnormal genome–lamina interactions in LMNA-deficient cardiomyocytes
Shen et al. demonstrate that LMNA reduction alters peripheral genome organization in human cardiomyocytes through cytoskeletal forces transmitted by the LI
📰 "Lamin A/C as a Molecular Link Between Nuclear Organization, Chromatin Dynamics, and Tumor Progression"
https://doi.org/doi:10.3390/cells15060501https://pubmed.ncbi.nlm.nih.gov/41892292/ #Lamin📰 "The cytoskeleton contributes to abnormal genome-lamina interactions in LMNA-deficient cardiomyocytes"
https://doi.org/doi:10.1083/jcb.202506137https://pubmed.ncbi.nlm.nih.gov/41891953/ #Cytoskeleton #Lamin📰 "A Conserved Geometric Code: Extracellular Matrix Curvature Directs Cell Migration Strategy via Nuclear Mechanosensing"
https://www.biorxiv.org/content/10.64898/2026.03.24.713851v1?rss=1 #Lamin
A Conserved Geometric Code: Extracellular Matrix Curvature Directs Cell Migration Strategy via Nuclear Mechanosensing
Cells navigate complex tissue microenvironments defined by intricate physical cues, yet how they interpret the three-dimensional geometry of the extracellular matrix (ECM) remains an open question. Current models often fail to account for the tortuous architectures found in physiological tissues. Here, we demonstrate that ECM curvature functions as a tissue-specific geometric code read by the cell nucleus. By mapping collagen architectures across cancers and tissues, we find unique curvature fingerprints preserved during metastasis. Using micro-engineered substrates, we show that high curvature imposes localized nuclear bending stress, triggering a Lamin A/C-cPLA2-Ca2+ mechanotransductive cascade. This sensor rewires the cytoskeleton from longitudinal stress fibers to a cortical actomyosin network, driving a sharp transition from fast mesenchymal migration to a slower, exploratory amoeboid phenotype. We term this "nuclear curvotaxis", establishing a physical principle linking static geometry to dynamic strategy, with implications for predicting metastatic risk, understanding immune exclusion, and designing bio-instructive scaffolds for tissue engineering.
### Competing Interest Statement
The authors have declared no competing interest.
National Natural Science Foundation of China, 12572352, 12225208, 12432015
Surface Project (Key Grant) of Chinese Medicine Education Assciation, 2024KTZ037
Technology Innovation Leading Program of Shaanxi, 2024QCY-KXJ069
Scientific Research Program Funded by Education Department of Shaanxi Provincial Government, 24JP160
📰 "A Case of Restrictive Dermopathy With Atypical Cardiac Anomalies and a Novel ZMPSTE24 Variant"
https://doi.org/doi:10.1002/ajmg.a.70149https://pubmed.ncbi.nlm.nih.gov/41877632/ #Lamin




