Studying the relationship between the size and the #anatomy and #physiology of #living organisms has proven useful in order to decipher what the key #physical constraints are that apply to them.
Here we consider both variations of #cellSize, using a drug that blocks mitosis without inhibiting growth, and of the interactions with the substrate, by considering the different stages of the quasi-static process of #cellSpreading, but also by using confinement, and thus presenting more than one substrate for the #cell to spread on. The fibroblasts used in this study present the archetypal shape of a cell spreading on a substrate, with a central cell body which, while flattening, retains a spherical cap shape, and is surrounded by a flat #lamella. We show that this shape is preserved isometrically between the cell size and cell confinement conditions. Moreover, the length ratio that characterises it establishes early during spreading, at the time when #focalAdhesion with the substrate develop. These #adhesion patches also collectively follow an isometric scaling with respect to cell spread area, in spite of having individually a shape, size and density which is insensitive to the global cell shape. We show that this isometry follows from the restriction of the distribution of focal adhesions to the flat lamella.
Since adhesion distribution is crucially important for maintaining the cell shape, we suggest that the observed allometry may be important for the cell to face the #mechanical challenges of physiological function.
Cell size homeostasis is tightly controlled throughout the cell cycle
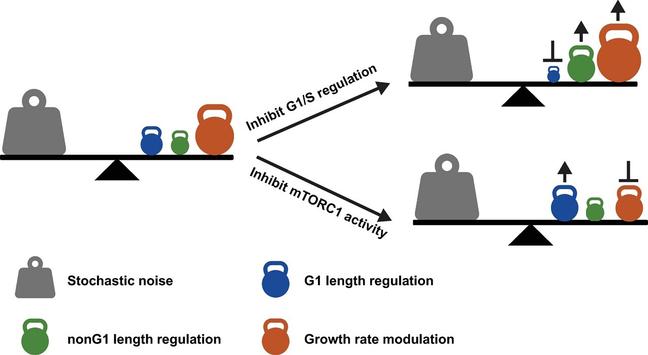
To achieve a stable size distribution over multiple generations, proliferating cells require a means of counteracting stochastic noise in the rate of growth, the time spent in various phases of the cell cycle, and the imprecision in the placement of the plane of cell division. In the most widely accepted model, cell size is thought to be regulated at the G1/S transition, such that cells smaller than a critical size pause at the end of G1 phase until they have accumulated mass to a predetermined size threshold, at which point the cells proceed through the rest of the cell cycle. However, a model, based solely on a specific size checkpoint at G1/S, cannot readily explain why cells with deficient G1/S control mechanisms are still able to maintain a very stable cell size distribution. Furthermore, such a model would not easily account for stochastic variation in cell size during the subsequent phases of the cell cycle, which cannot be anticipated at G1/S. To address such questions, we applied computationally enhanced quantitative phase microscopy (ceQPM) to populations of cultured human cell lines, which enables highly accurate measurement of cell dry mass of individual cells throughout the cell cycle. From these measurements, we have evaluated the factors that contribute to maintaining cell mass homeostasis at any point in the cell cycle. Our findings reveal that cell mass homeostasis is accurately maintained, despite disruptions to the normal G1/S machinery or perturbations in the rate of cell growth. Control of cell mass is generally not confined to regulation of the G1 length. Instead mass homeostasis is imposed throughout the cell cycle. In the cell lines examined, we find that the coefficient of variation (CV) in dry mass of cells in the population begins to decline well before the G1/S transition and continues to decline throughout S and G2 phases. Among the different cell types tested, the detailed response of cell growth rate to cell mass differs. However, in general, when it falls below that for exponential growth, the natural increase in the CV of cell mass is effectively constrained. We find that both mass-dependent cell cycle regulation and mass-dependent growth rate modulation contribute to reducing cell mass variation within the population. Through the interplay and coordination of these 2 processes, accurate cell mass homeostasis emerges. Such findings reveal previously unappreciated and very general principles of cell size control in proliferating cells. These same regulatory processes might also be operative in terminally differentiated cells. Further quantitative dynamical studies should lead to a better understanding of the underlying molecular mechanisms of cell size control.
Referenced link: https://phys.org/news/2023-03-cell-size-mechanism-cyanobacteria.html
Discuss on https://discu.eu/q/https://phys.org/news/2023-03-cell-size-mechanism-cyanobacteria.html
Originally posted by Phys.org / @physorg_com: http://nitter.platypush.tech/physorg_com/status/1639253255531405315#m
Researchers report novel #cellsize regulation mechanism in #cyanobacteria @PNASNews @PNASNexus https://pnas.org/doi/full/10.1073/pnas.2207963119 https://phys.org/news/2023-03-cell-size-mechanism-cyanobacteria.html
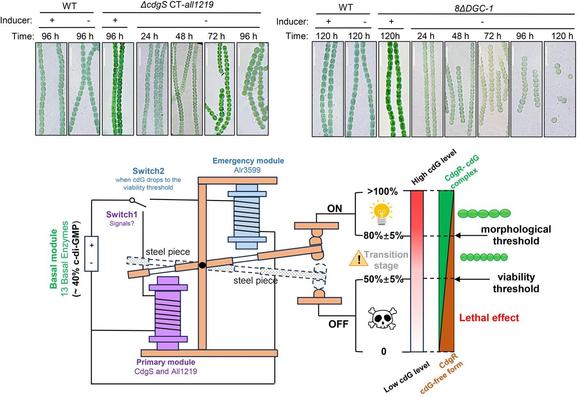
Researchers report novel cell size regulation mechanism in cyanobacteria
Cyanobacteria are the earliest known oxygenic photosynthetic organisms on Earth, and they played decisive roles in the evolution of the environment and the life on our planet. Cell morphology and cell size of different cyanobacteria species vary widely, but each species has inheritable and distinct cell morphology and cell size that are stably maintained through the generations. The underlying mechanisms of such a homeostasis have been unknown.