https://doi.org/doi:10.1073/pnas.2604349123
https://pubmed.ncbi.nlm.nih.gov/42201958/
#Mechanical #Actin #Force #Cell
the nautilus gadget
#Nautilus #Gadget #SteampunkStyle #Spiral #Futuristic #Mechanical #Artifact #Iridescent #Technology #SciFi #Glass #Brass #Steel
#Img2img #AiArt #AiArtists #AiArtCommunity #StableDiffusion
on things in your present life: https://aieris.art/featured/the-nautilus-gadget-eris-and-ai.html
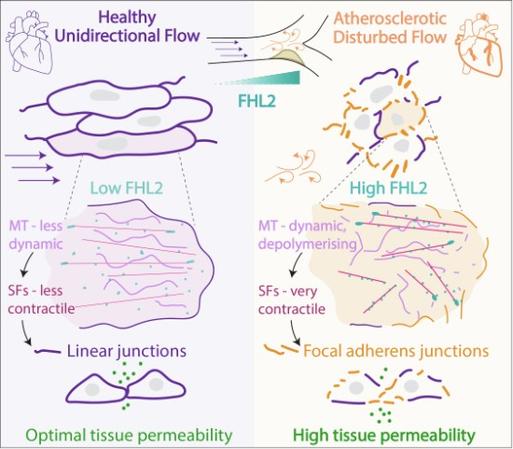
Endothelial tissues are essential mechanosensors in the vasculature, and defects in their response to mechanical cues such as blood flow can lead to endothelial dysfunction and cardiovascular diseases like atherosclerosis. Here, we explore how mechanoresponses tune endothelial tissue physiology and function. By bulk RNA sequencing in endothelial cells experiencing varying flow profiles, we identify a set of novel mechanosensitive genes associated with the cytoskeleton and adhesion structures. We focus on a cytoskeletal protein, Four-and-a-half LIM protein 2 (FHL2), which is consistently enriched in endothelial tissues experiencing atherosclerosis-prone disturbed flow, both in vitro and in vivo. We demonstrate that increased FHL2 expression is necessary and sufficient to induce hallmarks of atherosclerosis-like endothelial phenotypes, including aberrant cell morphology, discontinuous cell junctions, hypercontractility, and increased tissue permeability. Strikingly, this atherosclerosis-like phenotype requires the force-sensitive binding of FHL2 to the actin cytoskeleton. Mechanistically, we show that FHL2 controls endothelial tissue phenotypes by promoting RhoGTPase-dependent actomyosin contractility via release of the microtubule-bound RhoGTPase effector, GEF-H1. These findings reveal a positive mechanochemical feedback wherein FHL2 force-sensitivity tunes multi-scale mechanoresponses and endothelial tissue physiology.