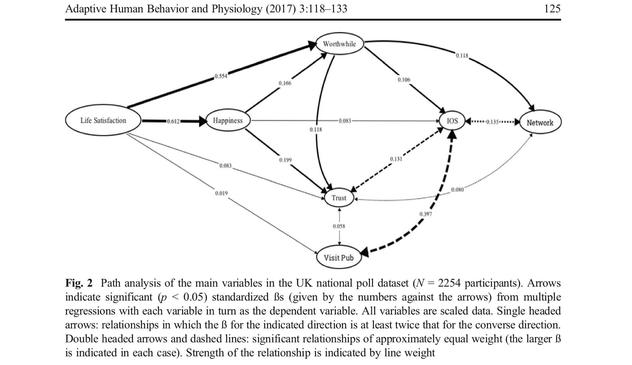
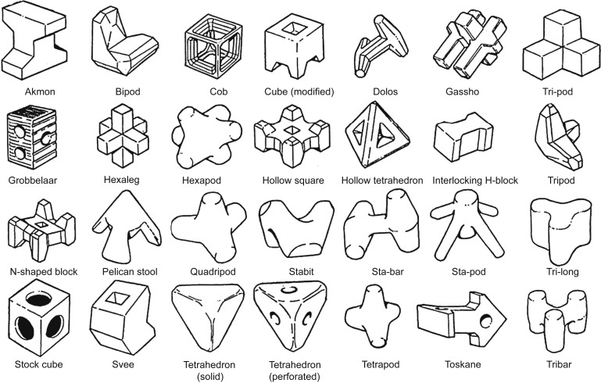
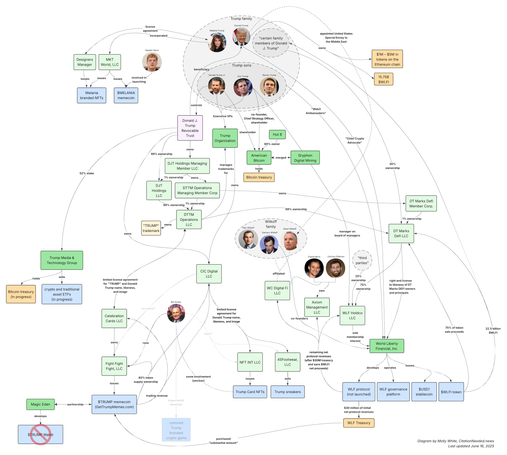
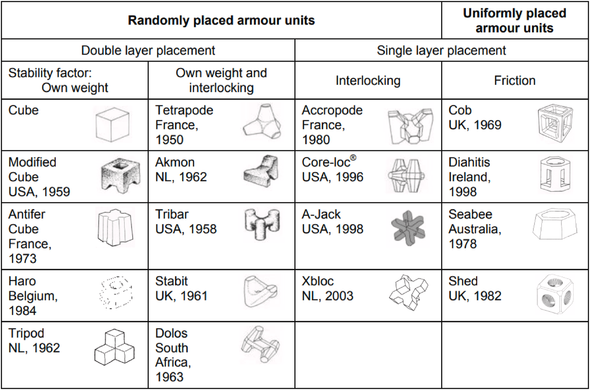
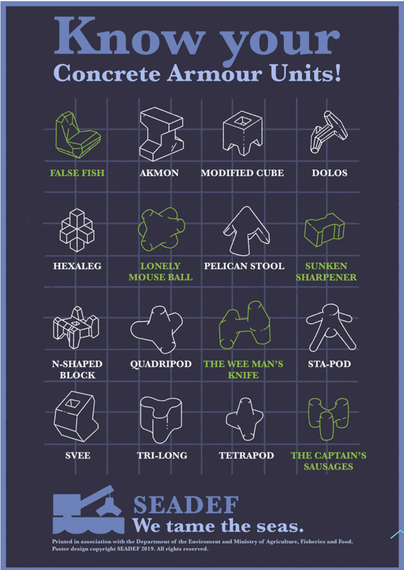
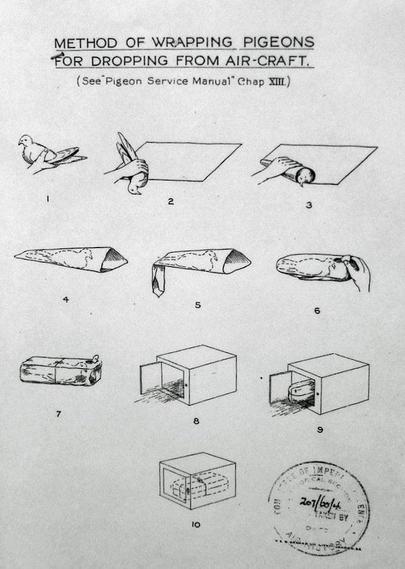
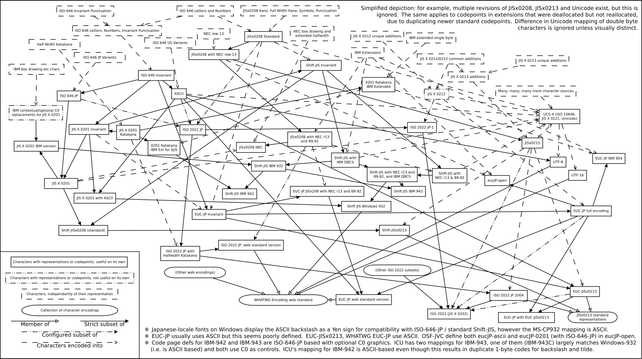
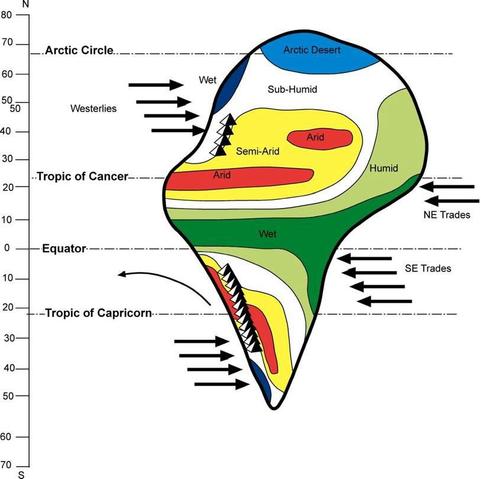
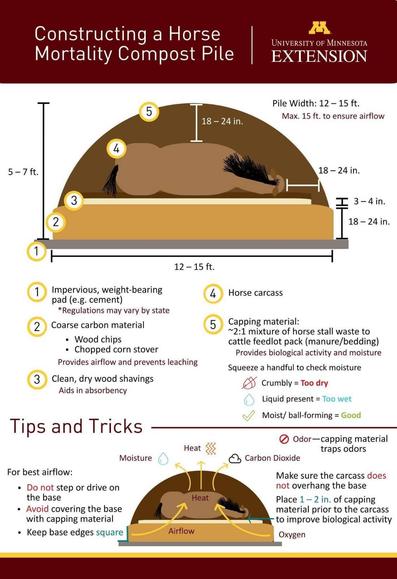
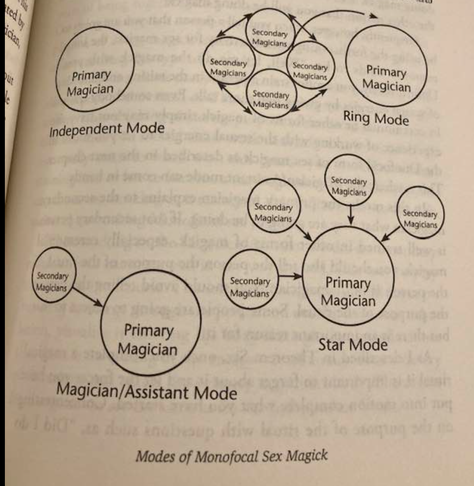
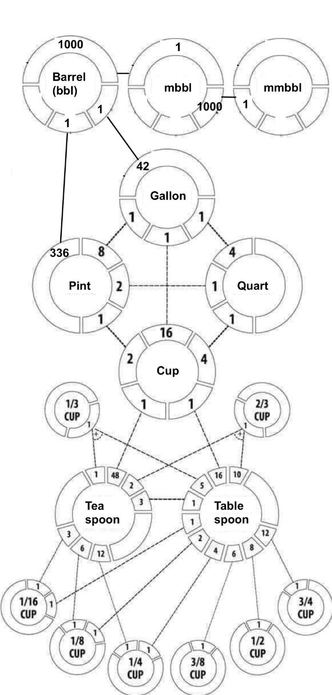
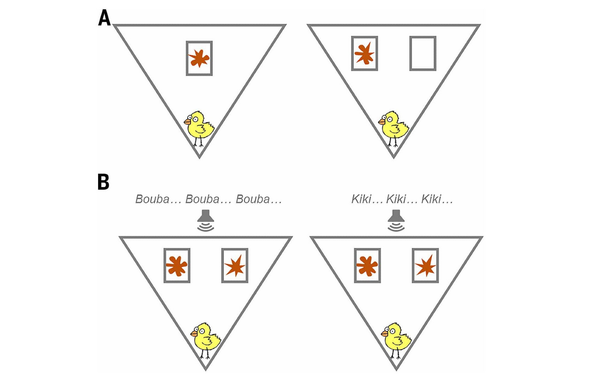
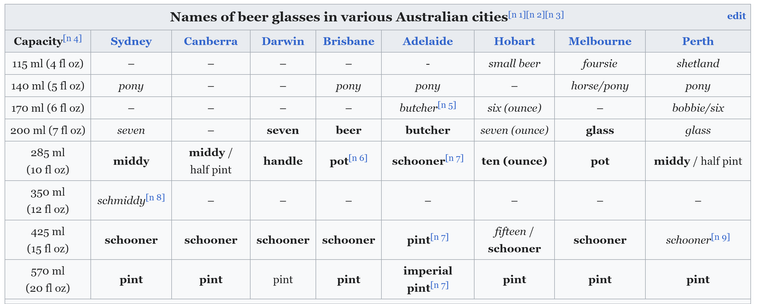
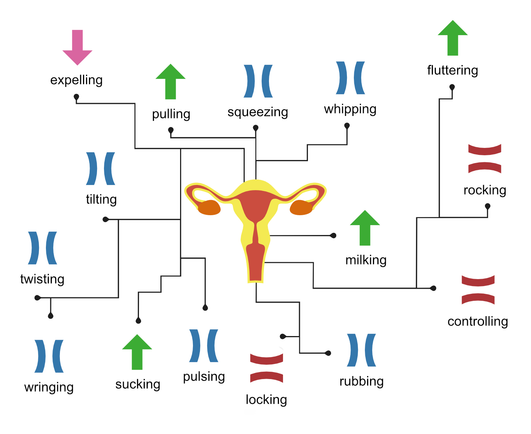
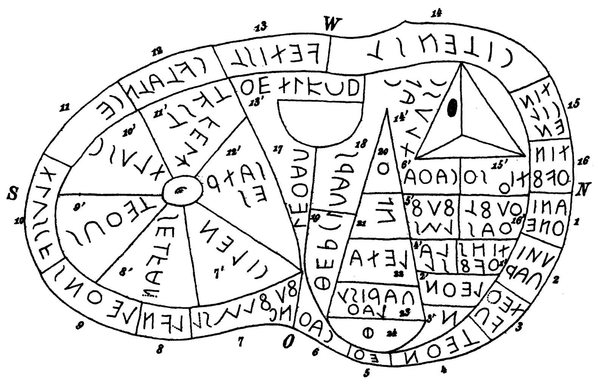
@llimllib
@hex @migurski @burritojustice @npseaver @yoz @jonty @nelson @peterme @v @jef @KevinMarks @sparks @mistertim @fil @air_pump @feralresearch @vruba @mizmay @randometc @meetar @stefpos @diagram @gsvoss @prehensile @inverse @shashashasha @ngsmcphrsn @zhenyi @jamescronin
@hex
@llimllib @migurski @burritojustice @npseaver @yoz @jonty @nelson @peterme @v @jef @KevinMarks @sparks @mistertim @fil @air_pump @feralresearch @vruba @mizmay @randometc @meetar @stefpos @diagram @gsvoss @prehensile @inverse @shashashasha @ngsmcphrsn @zhenyi @jamescronin

Matthew Dockrey (@[email protected])
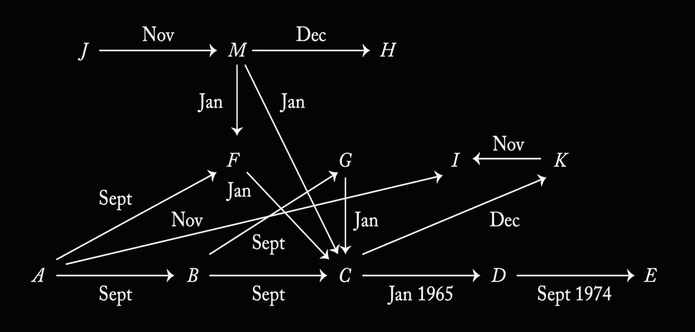
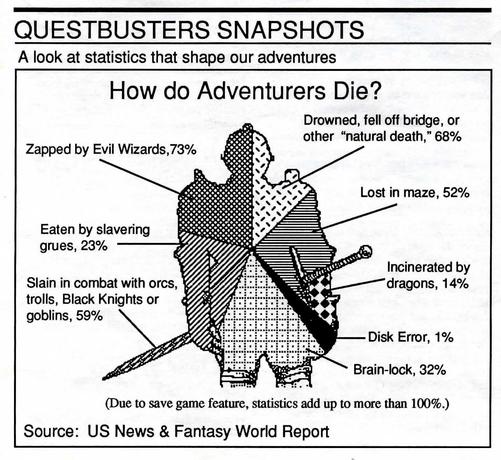
Attached: 1 image Scientific diagrams that look like shitposts is a fun genre, sure, but they've got nothing on epistemologists. (From Experience and Prediction by Hans Reichenbach.)
@mistertim @hex @migurski @llimllib @burritojustice @npseaver @yoz @jonty @peterme @v @jef @KevinMarks @sparks @fil @air_pump @feralresearch @vruba @mizmay @randometc @meetar @stefpos @diagram @gsvoss @prehensile @inverse @shashashasha @ngsmcphrsn @zhenyi @jamescronin
https://mathstodon.xyz/@johncarlosbaez/116381348796469355
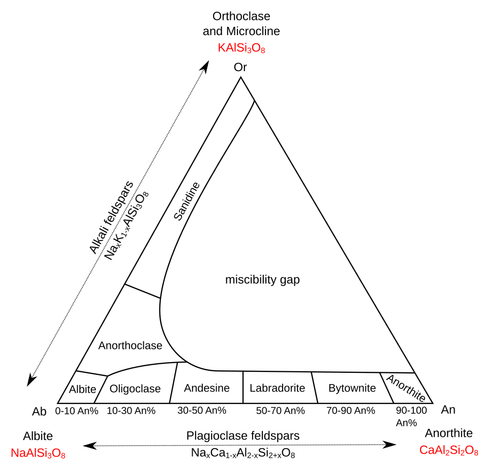
John Carlos Baez (@[email protected])
Attached: 1 image In the town of Quartzsite I picked up a beautiful chunk of labradorite. This mineral creates an eerie blue shimmer in the sunlight - a phenomenon called 'labradorescence'. Reading up on it, I discovered it's a form of feldspar. 60% of the Earth's crust is feldspar, and I know so little about this stuff! Turns out there are 3 fundamental kinds of feldspar: • orthoclase is potassium aluminosilicate • albite is sodium aluminosilicate • anorthite is calcium aluminosilicate Then there are lots of feldspars that contain different amounts of potassium, sodium and calcium. We get a triangle of feldspars with orthoclase, albite and anorthite at the corners. But not all points in this triangle are possible! There's a big region called the 'miscibility gap', where as you cool the molten mix it separates out! And there are also subtler problems. When you cool down the feldspar called labradorite, it separates out a little, forming tiny layers of two different kinds of stuff. When the thickness of these layers is the wavelength of visible light, you get a weird optical effect: labradorescence! (1/n)