`..phosphorylation of Optineurin by TBK1 induces the formation of filaments that phase separate upon binding to linear polyubiquitin.. the condensation of filamentous Optineurin with ubiquitylated cargo promotes the nucleation of cargo and its subsequent alignment with LC3-positive nascent autophagosomes, suggesting that co-condensation processes ensure directionality in selective autophagy`
https://advanced.onlinelibrary.wiley.com/doi/10.1002/advs.202509927
"These results have broad implications for biological processes regulated by membrane potential, particularly in contexts such as neuronal signaling, where condensate interactions with membranes may play a previously unrecognized regulatory role."
Phase separation of the PRPP amidotransferase into dynamic condensates promotes de novo purine synthesis in yeast
PRPP amidotransferase is the rate-limiting enzyme in de novo purine synthesis, but how its activity is regulated remains unclear. These authors show that PRPP forms intracellular condensates in response to high purine demand through phase separation, which is driven by TORC1-mediated macromolecular crowding of cytoplasm, facilitating de novo purine synthesis in yeast.
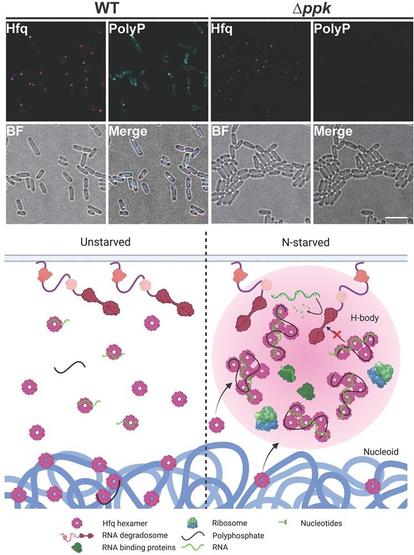
Synthetic compartments 'turbocharge' #bacteria for faster #protein production.
https://phys.org/news/2025-02-synthetic-compartments-turbocharge-bacteria-faster.html
Synthetic compartments 'turbocharge' bacteria for faster protein production
Biomedical engineers at Duke University have demonstrated a new synthetic approach that turbocharges bacteria into producing more of a specific protein, even proteins that would normally destroy them, such as antibiotics.
https://nachrichten.idw-online.de/2024/11/28/comeincell-launches-a-european-doctoral-network-to-train-the-next-generation-of-cell-scientists
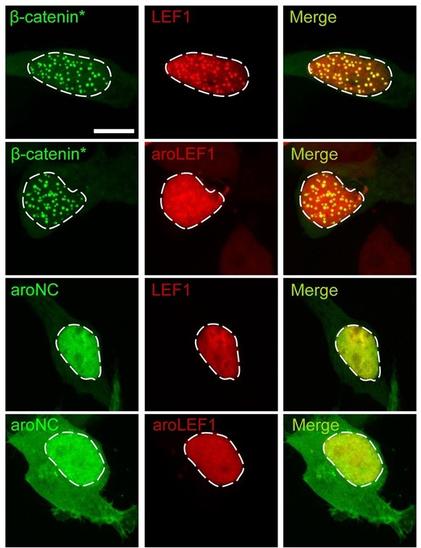
Wnt target gene activation requires β-catenin separation into biomolecular condensates
The Wnt/β-catenin signaling pathway plays numerous essential roles in animal development and tissue/stem cell maintenance. The activation of genes regulated by Wnt/β-catenin signaling requires the nuclear accumulation of β-catenin, a transcriptional co-activator. β-catenin is recruited to many Wnt-regulated enhancers through direct binding to T-cell factor/lymphoid enhancer factor (TCF/LEF) family transcription factors. β-catenin has previously been reported to form phase-separated biomolecular condensates (BMCs), which was implicated as a component of β-catenin’s mechanism of action. This function required aromatic amino acid residues in the intrinsically disordered regions (IDRs) at the N- and C-termini of the protein. In this report, we further explore a role for β-catenin BMCs in Wnt target gene regulation. We find that β-catenin BMCs are miscible with LEF1 BMCs in vitro and in cultured cells. We characterized a panel of β-catenin mutants with different combinations of aromatic residue mutations in human cell culture and Drosophila melanogaster. Our data support a model in which aromatic residues across both IDRs contribute to BMC formation and signaling activity. Although different Wnt targets have different sensitivities to loss of β-catenin’s aromatic residues, the activation of every target examined was compromised by aromatic substitution. These mutants are not defective in nuclear import or co-immunoprecipitation with several β-catenin binding partners. In addition, residues in the N-terminal IDR with no previously known role in signaling are clearly required for the activation of various Wnt readouts. Consistent with this, deletion of the N-terminal IDR results in a loss of signaling activity, which can be rescued by the addition of heterologous IDRs enriched in aromatic residues. Overall, our work supports a model in which the ability of β-catenin to form biomolecular condensates in the nucleus is tightly linked to its function as a transcriptional co-regulator.