Safety and immunogenicity of four sequential doses of NVX-CoV2373 in adults and adolescents: a phase 3, randomized, placebo-controlled trial (PREVENT-19)
The #RCT of #gigavaxx with #novavax (non-mRNA vaxx for #sarscov2 #covid19) shows sustained activity that partially extends to variants
https://www.medrxiv.org/content/10.1101/2024.11.07.24316930v1
Safety and immunogenicity of four sequential doses of NVX-CoV2373 in adults and adolescents: a phase 3, randomized, placebo-controlled trial (PREVENT-19)
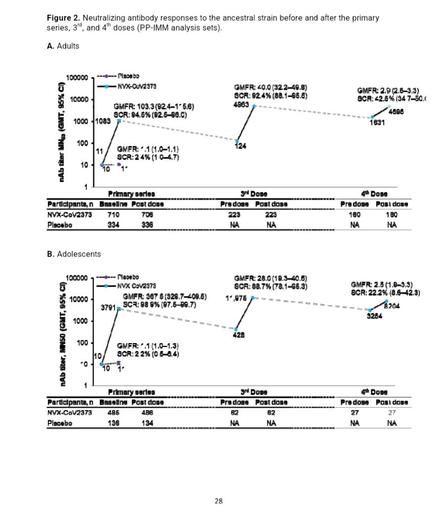
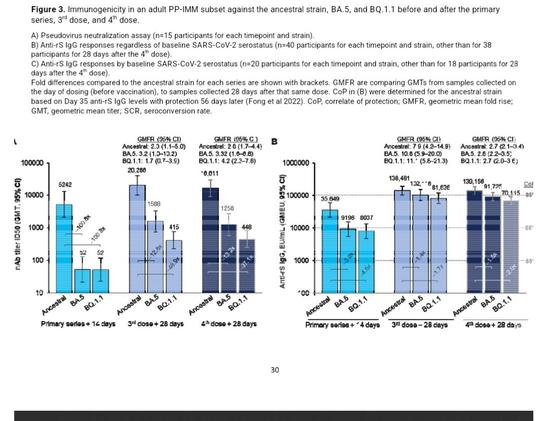
Background NVX-CoV2373, a recombinant SARS-CoV-2 spike (rS) protein vaccine with Matrix-M™ adjuvant, has been authorized for use in adults and adolescents. PREVENT-19 ([NCT04611802][1]/2019nCoV-301), a pivotal phase 3, randomized, placebo-controlled trial demonstrated robust efficacy of a primary, 2-dose series of NVX-CoV2373 against COVID-19. Methods Protocol expansions to PREVENT-19 included enrollment of adolescents (aged 12 to <18 years) and administration of 3rd and 4th doses of NVX-CoV2373 to adults and adolescents. Participants randomized 2:1 received NVX-CoV2373 or placebo 21 days apart; 3rd and 4th doses were administered ≥6 months after the preceding dose. Secondary and additional assessments included post-3rd- and 4th-dose immune responses (neutralizing antibody [nAb], anti-rS IgG, human angiotensin-converting enzyme-2-receptor binding inhibition [hACE2-RBI]) and response durability (post-3rd dose) to ancestral virus; cross-reactivity to Omicron subvariants; safety; and reactogenicity. Results Immune responses were observed against ancestral virus after two doses of NVX-CoV2373 but not after placebo. In both adults and adolescents, additional doses of NVX-CoV2373 increased nAb titers, anti-rS IgG levels, and hACE2-RBI; durable responses were recorded 8 months post 3rd dose. nAb responses post 3rd dose were noninferior to those post primary series. Cross-reactivity to BA.5 and BQ.1.1 variants was also observed, with anti-rS IgG levels post 3rd or 4th dose exceeding previously reported correlates of protection. Additional doses of NVX-CoV2373 were well tolerated, with no new safety signals. Conclusions NVX-CoV2373 elicited robust and durable humoral immune responses to ancestral SARS-CoV 2 as a 3rd and 4th dose after the primary series in adults and adolescents. Cross-reactivity to relevant variants provides insight into potential protection against antigenically related, but shifted, viral strains. Additional doses of NVX-CoV2373 were well tolerated with no new safety signals. These results support the utility of this vaccine platform and continued updates, based on currently circulating strains, to help effectively combat SARS-CoV-2 infection. ### Competing Interest Statement Karen L. Kotloff reports financial support was provided by Novavax, Inc. and the National Institute of Allergy and Infectious Diseases. Cynthia L. Gay reports financial support was provided by National Institute of Allergy and Infectious Diseases. Alice McGarry, Wayne Woo, Mingzhu Zhu, Shane Cloney-Clark, Joy Nelson, Haoua Dunbar, Miranda R. Cai, Iksung Cho, Zhaohui Cai, Raj Kalkeri, Joyce S. Plested, Nita Patel, Katherine Smith, Anthony M. Marchese, and Raburn M. Mallory report a relationship with Novavax, Inc. that includes: employment and equity or stocks. German Anez reports a relationship with Novavax, Inc that includes: employment (former) and equity or stocks. Gregory Glenn reports a relationship with Novavax, Inc. that includes: consulting or advisory, employment (former), and equity or stocks. Lisa Dunkle reports a relationship with Novavax, Inc. that includes: consulting or advisory, employment (former), and equity or stocks. Karen L. Kotloff reports a relationship with Novavax, Inc. that includes: funding grants. Medical writing support was funded by Novavax, Inc. If there are other authors, they declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. ### Clinical Trial NCT04611802 ### Funding Statement This work was supported by Novavax, Inc.; the Office of the Assistant Secretary for Preparedness and Response, Biomedical Advanced Research and Development Authority (BARDA; contract Operation Warp Speed: Novavax Project Agreement number 1 under Medical CBRN [Chemical, Biological, Radiological, and Nuclear] Defense Consortium base agreement no. 2020-530; Department of Defense no. W911QY20C0077); and the National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health. The NIAID provides grant funding to the HIV Vaccine Trials Network (HVTN) Leadership and Operations Center (UM1 AI68614), the HVTN Statistics and Data Management Center (UM1 AI68635), the HVTN Laboratory Center (UM1 AI68618), the HIV Prevention Trials Network Leadership and Operations Center (UM1 AI68619), the AIDS Clinical Trials Group Leadership and Operations Center (UM1 AI68636), and the Infectious Diseases Clinical Research Consortium leadership group (UM1 AI148684). ### Author Declarations I confirm all relevant ethical guidelines have been followed, and any necessary IRB and/or ethics committee approvals have been obtained. Yes The details of the IRB/oversight body that provided approval or exemption for the research described are given below: The following Institutional Review Boards / Independent Ethics committees reviewed and gave ethical approval for the PREVENT-19 study: Western Copernicus Group IRB, US; Great Plains IRB, US; Comite de etica en investigacion del Instituto Nacional de Ciencias Medicas y Nutricion, Salvador Zubiran, Mexico; Comite de etica en investigacion de la Unidad de Atencion Medica e Investigacion en Salud S.C., Mexico; Comite de etica en investigacion del Instituto Nacional de Salud Publica, Mexico; Comite de etica en investigacion de Medica Rio Mayo S.C., Mexico; Comite de etica en investigacion del Hospital La Mision S.A. de C.V., Mexico. All necessary patient/participant consent has been obtained and the appropriate institutional forms have been archived. I confirm that all necessary patient/participant consent has been obtained and the appropriate institutional forms have been archived, and that any patient/participant/sample identifiers included were not known to anyone (e.g., hospital staff, patients or participants themselves) outside the research group so cannot be used to identify individuals. Yes I understand that all clinical trials and any other prospective interventional studies must be registered with an ICMJE-approved registry, such as ClinicalTrials.gov. I confirm that any such study reported in the manuscript has been registered and the trial registration ID is provided (note: if posting a prospective study registered retrospectively, please provide a statement in the trial ID field explaining why the study was not registered in advance). Yes I have followed all appropriate research reporting guidelines, such as any relevant EQUATOR Network research reporting checklist(s) and other pertinent material, if applicable. Yes Study details can be found at https://clinicaltrials.gov/study/[NCT04611802][1]. Data that support the findings of this study are available in supplementary material and from the corresponding author upon reasonable request. [1]: /lookup/external-ref?link_type=CLINTRIALGOV&access_num=NCT04611802&atom=%2Fmedrxiv%2Fearly%2F2024%2F11%2F11%2F2024.11.07.24316930.atom