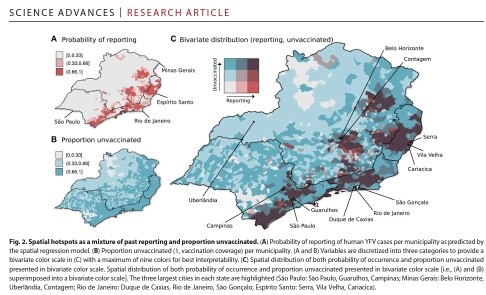
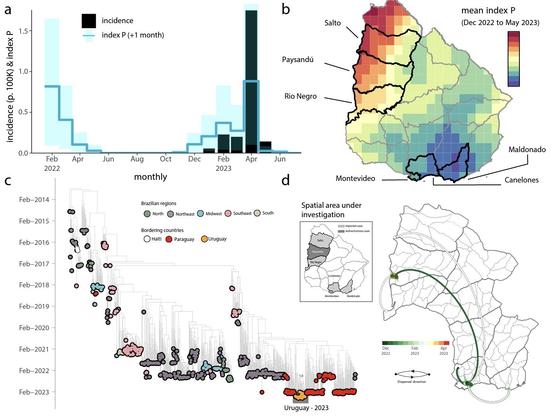
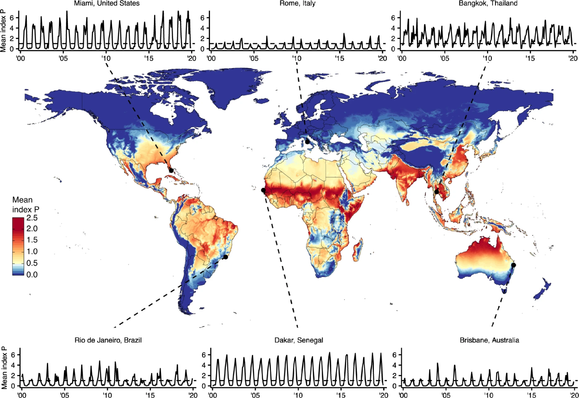
We reconstructed the recent transmission history of #YellowFeverVirus in #Brazil. A suitability index was able to capture the seasonality of #zoonotic cases. Spatial modeling revealed hotspots with both past reporting and low #vaccination coverage, coinciding with the largest urban centers in the Southeast. #Phylodynamics identified 3 lineages & provided proof of the directionality of a spatial corridor connecting the endemic North with the extra-Amazonian basin.
Spaceships & pathogens.
🇵🇹🇬🇧🇪🇺🌈🎮
| Scholar | https://scholar.google.com/citations?user=A4y9Yw8AAAAJ&hl=en |
| https://twitter.com/LourencoJML | |
| ORCID | https://orcid.org/0000-0002-9318-2581 |
| Católica Biomedical Research Center | https://ciencia.ucp.pt/pt/persons/jos%C3%A9-louren%C3%A7o |