For those interested, this screencast provides an overview of how the mutation spectrum can shape evo, in 3 modes (each with its own theory and empirical results)
* deleterious mut pressure
* ultimate source of raw materials
* introduction of novelty
https://www.youtube.com/watch?v=n8Tb2xatUNs
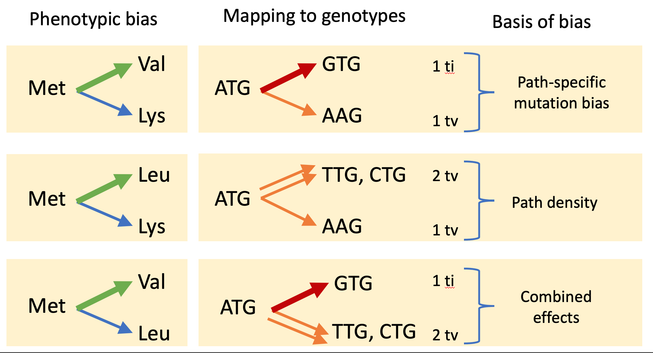
Most people will benefit from the intro video that explains some basic concepts, including the concept of a generative bias, i.e., a bias in the process of generating variation