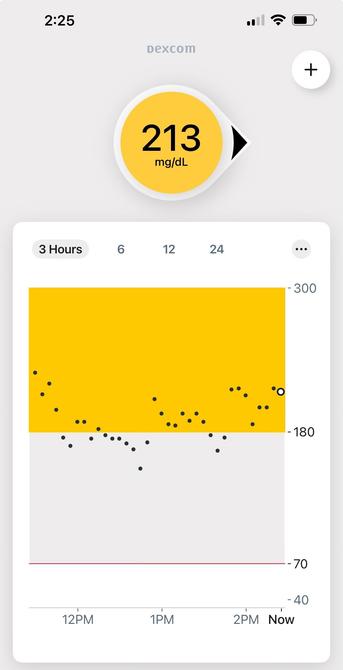
@VTDARKSIM did you presoak it (insert without activating it a day ahead of time)? It looks like components of the adhesive used on the G7 interfere and take about a day to disappear (hence the @^#%%^$#@ readings at the beginning of a session).
@cortig lol what?! Not saying that’s not true but it’s insane if it is and it’s gone to production that way.
@VTDARKSIM it got to production that way, and they got slapped on the wrist by the FDA for changing the formulation of the adhesive (coating) without approval and retesting. Some people claim they even did it despite internal memos warning that the new formulation interfered with readings (I don't have a good source for that).
https://www.medtechdive.com/news/dexcom-warning-letter-cgm-coating-change/743597/
@cortig hadn’t heard that, but this is my surprised face 😐