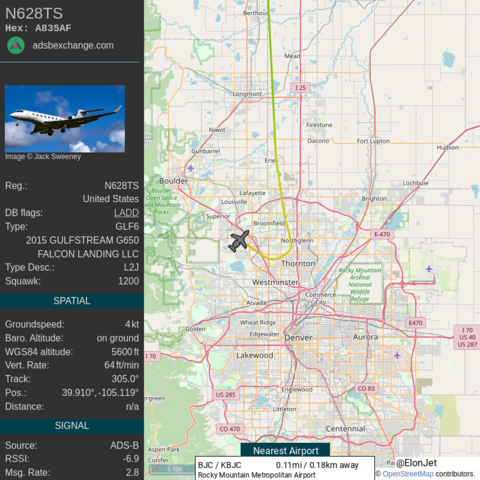
Flight Fuel Info ```
~ 868 gallons (3,287 liters).
~ 5,820 lbs (2,640 kg) of jet fuel used.
~ $5,514 cost of fuel.
~ 9 tons of CO2 emissions.```
So the CO2 emissions is roughly three times the fuel by weight. I'll never understand chemistry.
The fuel isn’t coal. There is some hydrogen in it as well which burns to produce water.
But at least I had the principle right of how you produce more CO2 than you had fuel 🙃
Oxygen is heavier than carbon, and two are attached to each carbon
The oxygen comes from the air
CO2 consists of three atoms. One carbon and two oxygens. the carbon comes from the fuel and the oxygen comes from the air and hasn't been in the fuel at all. So two thirds of the atoms come extra on top. The weight of carbon and oxygen is very roughly similar, so that in the end you have three times the amount of co2.
@rrrrrichard @elonjet Yes, the O2 comes from the air. The hydrocarbon molecules consist of H + C. The H is very light, the C is much heavier so dominates the weight of the fuel. C and O are near each other on the same row of the periodic table, so they weigh nearly the same.
So ignoring the H (which gets turned into water and not counted--the contrails you see) the majority of the fuel weight gets combined with twice as much in weight of oxygen (O2), so, yep, roughly 3x.
As an mere approximation:
the carbon (molucular weight 12) comes from the fuel and the oxygen (2 atoms with molucular weight 16=32) comes from the air.
Its not that simple but by that you know why there is more CO2 than fuel.
You could also say, the atmosphere loses two tons of oxygen with every ton of fossil fuel burnt.
Hope my English is understandable😉
Very understandable English. Thanks for this explanation. I hadn’t thought of it this way before.
C has an atomic mass of 12u, O has 16u, take away the Hs - > you end up roughly with factor 3.
@rrrrrichard @elonjet This is a common physics misunderstanding, rather than chemistry.
Kg/pounds/tons are a measure of mass, not weight. Gas is less dense than liquid, and jet fuel has a density of 3.16.
Weight is a force of attraction that two bodies in space have, so weight in physics is measured using Newtons.
The Oxygen(O) of the CO2 is added from the air.
@rrrrrichard @elonjet Yes, it's about 3 times the fuel weight used:
CO2 Emissions - Carbon Offset Guide
Carbon dioxide (CO2) is emitted during the combustion of kerosene jet fuel (referred to as ‘jet fuel’): 3.16 kg CO2 are emitted per kilogram of jet fuel combusted (ICAO, 2017). The CO2 emissions during the production of kerosene (including transport...
I'm no chemistry wizard either. But this one is fairly simple. Jet fuel contains about 80 percent carbon. In the combustion process oxygen from the air is added. The oxygen weighs roughly 3 times as much as the carbon, hence the higher weight.
The fuel is made of hydrocarbons, which are carbon chains with a bunch of hydrogens attached to them. The fuel is mixed with oxygen and burned, and the hydrocarbons are torn apart.
Two hydrogen atoms combine with one oxygen atom to form water, H2O. Each carbon combines with two oxygens to form carbon dioxide, CO2.
Now to explain the weight difference, we have to introduce the unit of moles. (1/2)
One mole is 6.022 x 10^23 atoms. (This number is called Avogadro’s number.) One mole of carbon atoms weighs 12.01g, a mole of oxygen atoms weighs 16.00g, and a mole of hydrogen atoms weighs only 1.01g.
Assuming each carbon in a hydrocarbon is one “link” and has two hydrogens attached, each mole of “links” in a hydrocarbon chain weighs 12.01 + (2)1.01 = 14.03g. When burned, the carbon becomes CO2: 12.01 + 2(16.00) = 44.01g of CO2. (2/3)
That’s 44.01g CO2 per 14.03g of hydrocarbon, which is approximately 3.137 times as much by mass.
I hope this explanation was clear, let me know if you have questions.
(3/3) (yes I thought this would only take 2)
Thank you for this interesting service.