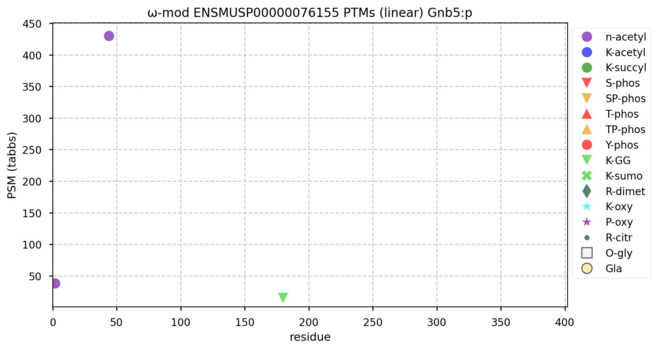
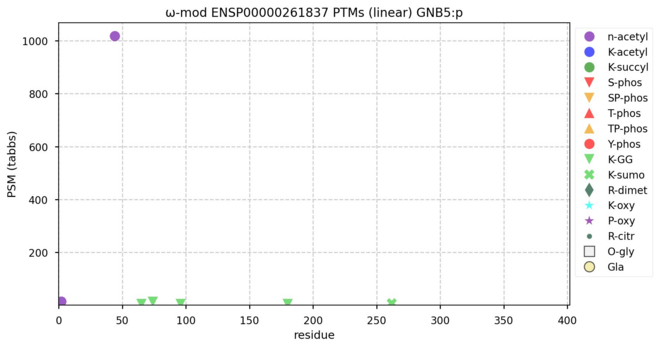
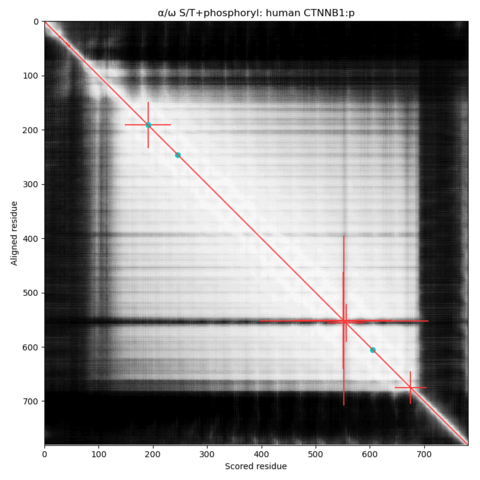
https://www.clubhouse.com/invite/1djuwJ4Vryd67KE608qDDzqKzwWIZ85XYd:cSGta-n3TL7ldlndrmNr5ld8d4YBmt6eECfXak3tS-U hey - you should join us Jan 31, 10:00 am for Proteomics Old-Time Radio Hour.
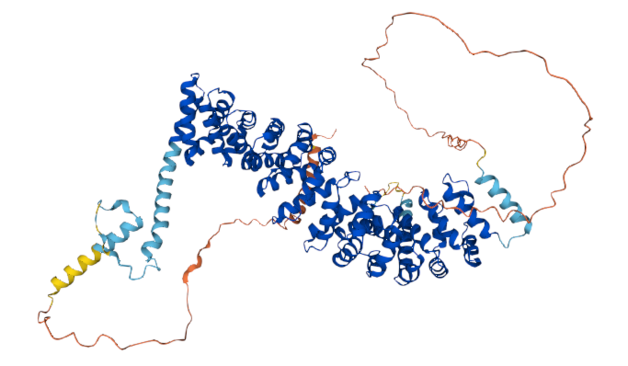
Man why do they always use my pic and not you @benneely.com 🤣 #proteomics #teamProteome