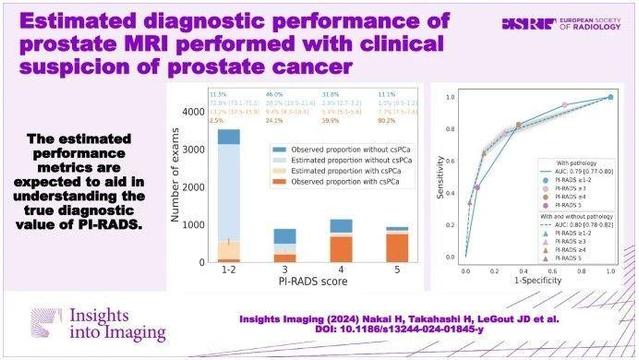
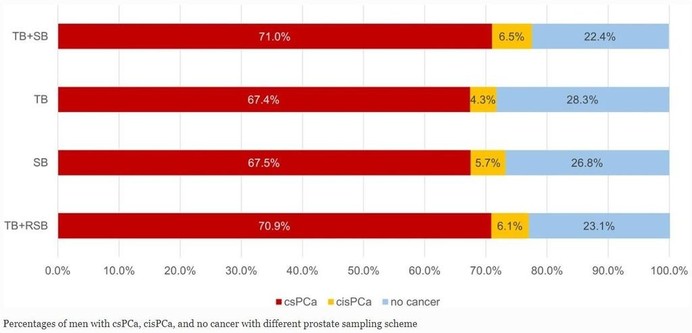
This retrospective study assessed the diagnostic performance of prostate MRI by estimating the proportion of clinically significant #ProstateCancer (csPCa) in patients without prostate pathology. It found varying csPCa proportions based on PI-RADS scores, with sensitivity of 76.6–77.3%, specificity of 67.5–78.6%, and NPV of 84.4–87.2%. (Hirotsugu Nakai et al.)
Estimated diagnostic performance of prostate MRI performed with clinical suspicion of prostate cancer - Insights into Imaging
Purpose To assess the diagnostic performance of prostate MRI by estimating the proportion of clinically significant prostate cancer (csPCa) in patients without prostate pathology. Materials and methods This three-center retrospective study included prostate MRI examinations performed for clinical suspicion of csPCa (Grade group ≥ 2) between 2018 and 2022. Examinations were divided into two groups: pathological diagnosis within 1 year after the MRI (post-MRI pathology) is present and absent. Risk prediction models were developed using the extracted eleven common predictive variables from the patients with post-MRI pathology. Then, the csPCa proportion in the patients without post-MRI pathology was estimated by applying the model. The area under the receiver operating characteristic curve (AUC), sensitivity, specificity, and positive and negative predictive values (PPV/NPV) of prostate MRI in diagnosing csPCa were subsequently calculated for patients with and without post-MRI prostate pathology (estimated statistics) with a positive threshold of PI-RADS ≥ 3. Results Of 12,191 examinations enrolled (mean age, 65.7 years ± 8.4 [standard deviation]), PI-RADS 1–2 was most frequently assigned (55.4%) with the lowest pathological confirmation rate of 14.0–18.2%. Post-MRI prostate pathology was found in 5670 (46.5%) examinations. The estimated csPCa proportions across facilities were 12.6–15.3%, 18.4–31.4%, 45.7–69.9%, and 75.4–88.3% in PI-RADS scores of 1–2, 3, 4, and 5, respectively. The estimated (observed) performance statistics were as follows: AUC, 0.78–0.81 (0.76–0.79); sensitivity, 76.6–77.3%; specificity, 67.5–78.6%; PPV, 49.8–66.6% (52.0–67.7%); and NPV, 84.4–87.2% (82.4–86.6%). Conclusion We proposed a method to estimate the probabilities harboring csPCa for patients who underwent prostate MRI examinations, which allows us to understand the PI-RADS diagnostic performance with several metrics. Clinical relevance statement The reported estimated performance metrics are expected to aid in understanding the true diagnostic value of PI-RADS in the entire prostate MRI population performed with clinical suspicion of prostate cancer. Key Points Calculating performance metrics only from patients who underwent prostate biopsy may be biased due to biopsy selection criteria, especially in PI-RADS 1–2. The estimated area under the receiver operating characteristic curve of PI-RADS in the entire prostate MRI population ranged from 0.78 to 0.81 at three facilities. The estimated statistics are expected to help us understand the true PI-RADS performance and serve as a reference for future studies. Graphical Abstract