https://www.the-scientist.com/cancer-cells-give-orders-71792 #cancer #fibroblasts #progression #CAF #EndothelialCells
A novel prognostic signature and immune microenvironment characteristics associated with disulfidptosis in papillary thyroid carcinoma based on single-cell RNA sequencing
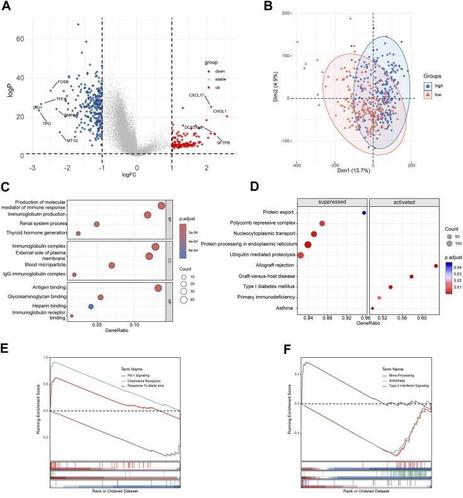
Background: Disulfidptosis is a newly discovered form of regulated cell death. The research on disulfidptosis and tumor progression remains unclear. Our research aims to explore the relationship between disulfidptosis-related genes (DRGs) and the clinical outcomes of papillary thyroid carcinoma (PTC), and its interaction on the tumor microenvironment.Methods: The single-cell RNA seq data of PTC was collected from GEO dataset GSE191288. We illustrated the expression patterns of disulfidptosis-related genes in different cellular components in thyroid cancer. LASSO analyses were performed to construct a disulfidptosis associated risk model in TCGA-THCA database. GO and KEGG analyses were used for functional analyses. CIBERSORT and ESTIMATE algorithm helped with the immune infiltration estimation. qRT‒PCR and flow cytometry was performed to validate the hub gene expression and immune infiltration in clinical samples.Results: We clustered PTC scRNA seq data into 8 annotated cell types. With further DRGs based scoring analyses, we found endothelial cells exhibited the most relationship with disulfidptosis. A 4-gene risk model was established based on the expression pattern of DRGs related endothelial cell subset. The risk model showed good independent prognostic value in both training and validation dataset. Functional enrichment and genomic feature analysis exhibited the significant correlation between tumor immune infiltration and the signature. The results of flow cytometry a...
Coronary #EndothelialCells want WT1 as well
A Research Highlight showcasing new work from Marina Ramiro-Pareta, Ofelia Martínez-Estrada & colleagues
Read the full #OpenAccess Research Article, 'Endothelial deletion of #Wt1 disrupts coronary #angiogenesis and #myocardium development', here:
https://journals.biologists.com/dev/article/150/6/dev201147/297331/Endothelial-deletion-of-Wt1-disrupts-coronary
We interviewed the authors, Marina Ramiro-Pareta & Ofelia Martínez-Estrada for our 'The people behind the papers' series:
https://journals.biologists.com/dev/article/150/6/dev201740/297330/The-people-behind-the-papers-Marina-Ramiro-Pareta
Coronary endothelial cells want WT1 as well!
WT1 is a transcription factor expressed in the heart epicardium (the outermost layer of the heart) and is required for epicardial development, with Wt1 knockout mice displaying cardiovascular defects and embryonic lethality. Although WT1 is also expressed in coronary endothelial cells (ECs), little was known about its role in these cells and its contribution to blood vessel formation. Now, Ofelia Martinez-Estrada and colleagues develop an inducible knockout mouse line (Wt1KOΔEC) to specifically knockout Wt1 in Pdgfb-expressing coronary ECs starting between E11.5 and E13.5. The authors show that Wt1KOΔEC mice have impaired coronary blood vessel and myocardium (heart muscle) development, caused by a reduction in proliferating cardiomyocytes and ECs. To understand these defects, the researchers transcriptionally profile Wt1KOΔEC coronary ECs and reveal that WT1 is a major regulator of their transcriptome, with mutant ECs failing to upregulate key maturation genes. In addition, using immunostaining, image analysis and in vitro approaches, they show that deletion of Wt1 impairs coronary EC proliferation, migration and maturation. Loss of Wt1 also affects the differentiation into venous and arterial ECs in early coronary plexus formation and coronary artery formation in later development. Together, these data demonstrate that WT1 is crucial for coronary EC development.
Take a look at this amusing repair of 2-photon vessel injury by 2 adjacent #EndothelialCells
Dr Karen Hirschi & Valentina Greco labs bioRxiv 2022
https://www.biorxiv.org/content/10.1101/2022.11.02.514907v1.full
Check out our recent paper on #vascular cell apoptotic vulnerabilities to #cancer therapeutics, including #radiotherapy, #chemotherapy and targeted agents (e.g. #proteasome inhibitors, #BH3mimetics). We find that #EndothelialCells remain primed for #apoptosis throughout development into adulthood and may represent a “weakest link” vulnerability in multiple tissues for development of toxicities. https://www.science.org/doi/full/10.1126/sciadv.abn6579
#Celldeath #CardioOncology #VigiBase #RadioBiology #BH3Profiling
You were close, but none of those !
These are #EndothelialCells 👍🏼
Referenced link: https://medicalxpress.com/news/2022-10-corrupt-endothelial-cells-blood-cancer.html
Discuss on https://discu.eu/q/https://medicalxpress.com/news/2022-10-corrupt-endothelial-cells-blood-cancer.html
Originally posted by Phys.org / @[email protected]: https://twitter.com/medical_xpress/status/1582803928995201025#m
RT by @physorg_com: Corrupt #endothelialcells found to protect blood #cancer cells from chemotherapy @WeillCornell https://ashpublications.org/blood/article/doi/10.1182/blood.2022015414/486319/Endothelial-leukemia-interactions-remodel-drug https://medicalxpress.com/news/2022-10-corrupt-endothelial-cells-blood-cancer.html
Corrupt endothelial cells found to protect blood cancer cells from chemotherapy
Endothelial cells—the cells that line blood vessels—grown alongside leukemia cells become corrupted and rescue the cancer cells from many chemotherapy drugs, a study by Weill Cornell Medicine investigators found.