Martin Posch
- 47 Followers
- 89 Following
- 44 Posts
An LLM "creates textual claims, and then predicts the citations that might be associated with similar text. Obviously, this practice violates all norms of scholarly citation.
At best, LLMs gesticulate toward the shoulders of giants."
@emilymbender , Jevin West, and I contributed to this perspective piece in PNAS. We took a skeptical position; others are very much enthusiasts. Before you pillory me for some random quote in this article, we strongly disagree with some of the claims in the other perspectives.
https://science.orf.at/stories/3228390/
Das Wort "Fediverse" in einem Artikel vom ORF,... und noch nichtmal falsch verwendet oder so.
Too little, too late, aber trotzdem irgendwie schön.
Kommunikation: Über 60 Unis und Forschungsinstitute verlassen X
Mehr als 60 deutschsprachige Hochschulen und Forschungsinstitutionen haben ihren Ausstieg beim Kurzbotschaftendienst X angekündigt. Sie begründeten dies in einer gemeinsamen Erklärung damit, dass die Ausrichtung der von US-Milliardär Elon Musk kontrollierten Plattform mit ihren Grundwerten wie „Weltoffenheit, wissenschaftliche Integrität, Transparenz und demokratischer Diskurs“ nicht vereinbar sei.
Stratification in Randomised Clinical Trials and Analysis of Covariance: Some Simple Theory and Recommendations
A simple device for balancing for a continuous covariate in clinical trials is to stratify by whether the covariate is above or below some target value, typically the predicted median. This raises an issue as to which model should be used for modelling the effect of treatment on the outcome variable, $Y$. Should one fit, the stratum indicator, $S$, the continuous covariate, $X$, both or neither? This question has sometimes been investigated using simulations targeting the overall effect on inferences about treatment, in terms, for example, of power for a given alternative hypothesis. However, when a covariate is added to a linear model there are three consequences for inference: 1) the mean square error effect, 2) the variance inflation factor and 3) second order precision. We consider that it is valuable to consider these three factors separately, even if, ultimately, it is their joint effect that matters. We present some simple theory, concentrating in particular on the variance inflation factor, that may be used to guide trialists in their choice of model. We also consider the case where the precise form of the relationship between the outcome and the covariate is not known. We conclude by recommending that the continuous covariate should always be in the model but that, depending on circumstances, there may be some justification in fitting the stratum indicator also.
New preprint/report:
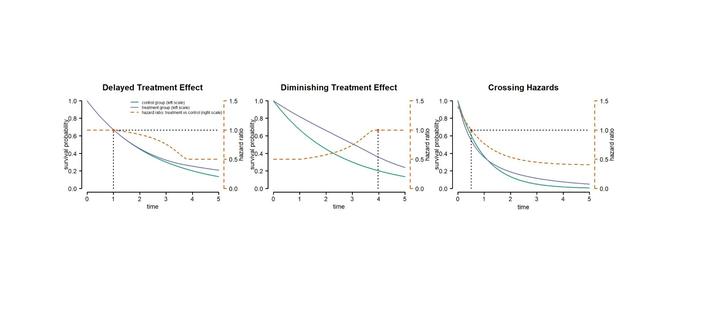
"A Review of EMA Public Assessment Reports where Non-Proportional Hazards were Identified"
A Review of EMA Public Assessment Reports where Non-Proportional Hazards were Identified
While well-established methods for time-to-event data are available when the proportional hazards assumption holds, there is no consensus on the best approach under non-proportional hazards. A wide range of parametric and non-parametric methods for testing and estimation in this scenario have been proposed. In this review we identified EMA marketing authorization procedures where non-proportional hazards were raised as a potential issue in the risk-benefit assessment and extract relevant information on trial design and results reported in the corresponding European Assessment Reports (EPARs) available in the database at paediatricdata.eu. We identified 16 Marketing authorization procedures, reporting results on a total of 18 trials. Most procedures covered the authorization of treatments from the oncology domain. For the majority of trials NPH issues were related to a suspected delayed treatment effect, or different treatment effects in known subgroups. Issues related to censoring, or treatment switching were also identified. For most of the trials the primary analysis was performed using conventional methods assuming proportional hazards, even if NPH was anticipated. Differential treatment effects were addressed using stratification and delayed treatment effect considered for sample size planning. Even though, not considered in the primary analysis, some procedures reported extensive sensitivity analyses and model diagnostics evaluating the proportional hazards assumption. For a few procedures methods addressing NPH (e.g.~weighted log-rank tests) were used in the primary analysis. We extracted estimates of the median survival, hazard ratios, and time of survival curve separation. In addition, we digitized the KM curves to reconstruct close to individual patient level data. Extracted outcomes served as the basis for a simulation study of methods for time to event analysis under NPH.
My thesis is finally not only handed in and graded but also in our universities thesis repository 🎉
Our paper "Methods for nonproportional hazards in clinical trials: A systematic review" has been published 🎉
Many thanks to the main authors: @huber8 @bardo1
❓ What methods are available for non-proportional hazards in clinical trials?
Our #SystematicReview is now available on arXiv
➡️https://arxiv.org/abs/2306.16858
Contributors include @bardo1 @stats_tobby @poschm @friede1
Methods for non-proportional hazards in clinical trials: A systematic review
For the analysis of time-to-event data, frequently used methods such as the log-rank test or the Cox proportional hazards model are based on the proportional hazards assumption, which is often debatable. Although a wide range of parametric and non-parametric methods for non-proportional hazards (NPH) has been proposed, there is no consensus on the best approaches. To close this gap, we conducted a systematic literature search to identify statistical methods and software appropriate under NPH. Our literature search identified 907 abstracts, out of which we included 211 articles, mostly methodological ones. Review articles and applications were less frequently identified. The articles discuss effect measures, effect estimation and regression approaches, hypothesis tests, and sample size calculation approaches, which are often tailored to specific NPH situations. Using a unified notation, we provide an overview of methods available. Furthermore, we derive some guidance from the identified articles. We summarized the contents from the literature review in a concise way in the main text and provide more detailed explanations in the supplement.