FDA grants breakthrough device designation to ReVision Implant visual cortex prosthesis - Med-Tech Insights
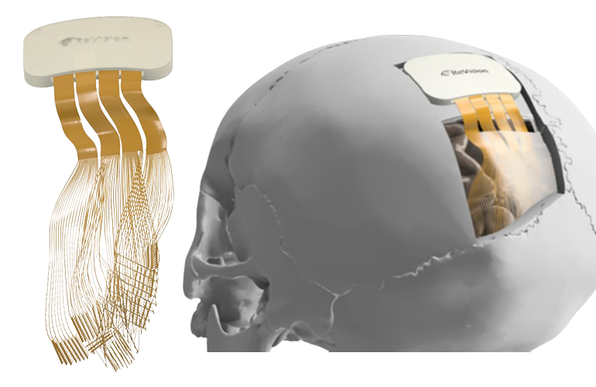
Belgian neurotechnology startup ReVision Implant has received Breakthrough Device designation from the U.S. Food and Drug Administration (FDA) for its visual cortical prosthesis, Occular, clearing the way toward first-in-human clinical trials. The Occular system is designed to restore functional vision in people with severe blindness, including patients whose condition cannot be treated with retinal implants or optic nerve therapies.