@kitten_tech - I don't know anything else like water. Its charge asymmetry and lightness are important. Some unusual properties, from Khan Academy:
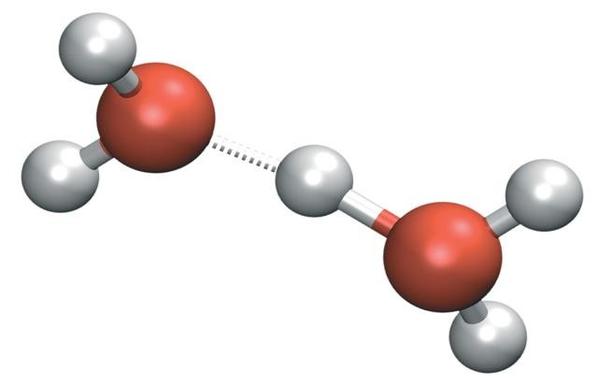
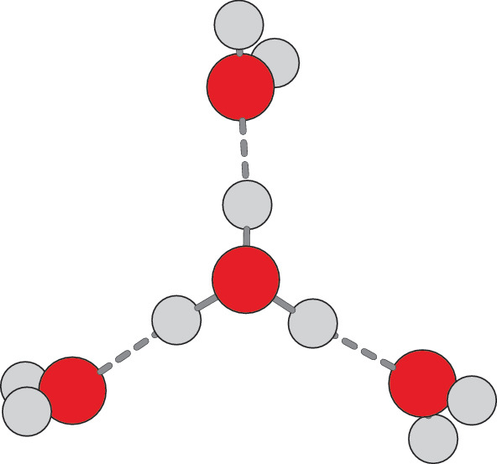
• Water is polar. Water molecules are polar, with partial positive charges on the hydrogens, a partial negative charge on the oxygen, and a bent overall structure. This is because oxygen is more electronegative, meaning that it is better than hydrogen at attracting electrons.
• Water is an excellent solvent. Water has the unique ability to dissolve many polar and ionic substances.
• Water has high heat capacity.
• Water has high heat of vaporization.
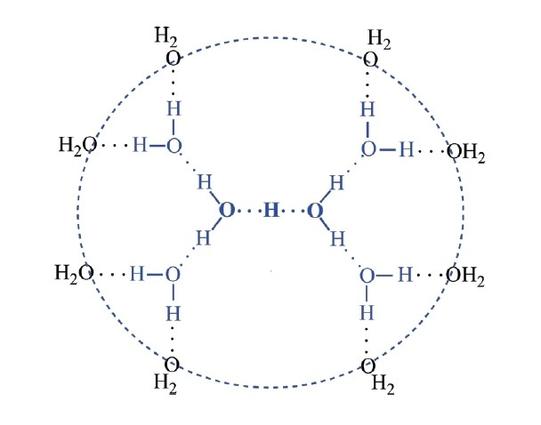
• Water has cohesive and adhesive properties. Water molecules have strong cohesive forces due to their ability to form hydrogen bonds with one another. Cohesive forces are responsible for surface tension, the tendency of a liquid’s surface to resist rupture when placed under tension or stress. Water also has adhesive properties that allow it to stick to substances other than itself.
• Water is less dense as a solid than as a liquid. As water freezes, the molecules form a crystalline structure that spaces the molecules further apart than in liquid water.
I'll add that it has a high latent heat - the heat required to melt it.
https://www.khanacademy.org/science/ap-biology/chemistry-of-life/structure-of-water-and-hydrogen-bonding/a/hs-water-and-life-review
@enoch_exe_inc