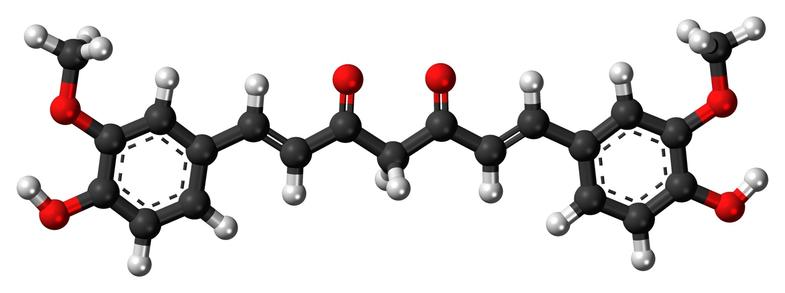
Suppose you were trying to invent a bright orange powder that could easily dye clothes and be hard to wash off. Using your knowledge of quantum mechanics you'd design this symmetrical molecule where an electron's wavefunction can vibrate back and forth along a chain of carbons at the frequency of green light. Absorbing green light makes it look orange! And this molecule doesn't dissolve in water.
Yes: you'd invent turmeric!
Or more precisely 'curcurmin', the molecule that gives turmeric its special properties.
The black atoms are carbons, the white are hydrogens and the red are oxygens.
Read on and check out what pure curcurmin looks like.
(1/n)