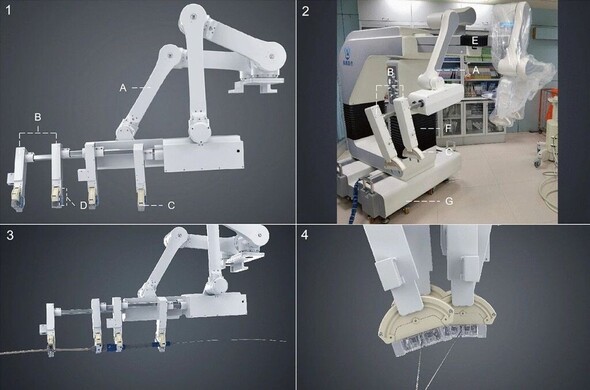
A novel endovascular #robotic-assisted system for endovascular #aortic repair demonstrates practicability, safety, and procedural effectiveness comparable to manual operation. (Chao Song et al.)
🔗 https://link.springer.com/article/10.1007/s00330-023-09810-x
A novel endovascular robotic-assisted system for endovascular aortic repair: first-in-human evaluation of practicability and safety - European Radiology
Objectives To assess the practicability and safety of a novel endovascular robotic system for performing endovascular aortic repair in human. Methods A prospective observational study was conducted in 2021 with 6 months post-operative follow-up. Patients with aortic aneurysms and clinical indications for elective endovascular aortic repair were enrolled in the study. The novel developed robotic system is applicable for the majority of commercial devices and various types of endovascular surgeries. The primary endpoint was technical success without in-hospital major adverse events. Technical success was defined as the ability of the robotic system to complete all procedural steps based on procedural segments. Results The first-in-human evaluation of robot-assisted endovascular aortic repair was performed in five patients. The primary endpoint was achieved in all patients (100%). There were no device- or procedure-related complications or no in-hospital major adverse events. The operation time and total blood loss in these cases were equal to those in the manual procedures. The radiation exposure of the surgeon was 96.5% lower than that in the traditional position while the radiation exposure of the patients was not significantly increased. Conclusions Early clinical evaluation of the novel endovascular aortic repair in endovascular aortic repair demonstrated practicability, safety, and procedural effectiveness comparable to manual operation. In addition, the total radiation exposure of the operator was significantly lower than that of traditional procedures. Clinical relevance statement This study applies a novel approach to perform the endovascular aortic repair in a more accurate and minimal-invasive way and lays the foundation for the perspective automation of the endovascular robotic system, which reflects a new paradigm for endovascular surgery. Key Points • This study is a first-in-human evaluation of a novel endovascular robotic system for endovascular aortic repair (EVAR). • Our system might reduce the occupational risks associated with manual EVAR and contribute to achieving a higher degree of precision and control. • Early evaluation of the endovascular robotic system demonstrated practicability, safety, and procedural effectiveness comparable to that of manual operation.
Explore our recent issues and back issues covering endovascular procedures from head to toe: https://evtoday.com/archive/2023
Images showcase several of our recent 2023 issue topics. #endovascular #vascularsurgery #interventionalradiology #irad #teamwork #stroke #aortic #embolization
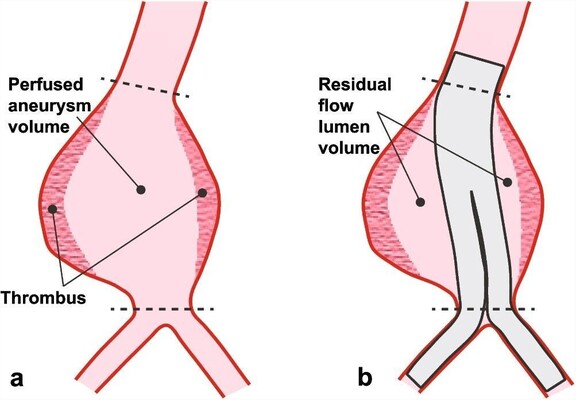
Shape memory polymer (SMP) devices for #aortic #aneurysm sac embolization during endovascular repair 🔧 shown to be feasible and safe. (Alexander Massmann et al.)
#EuropeanRadiologyExperimental #OpenAccess
🔗 https://eurradiolexp.springeropen.com/articles/10.1186/s41747-023-00328-x
Feasibility of aortic aneurysm sac embolization using a novel shape memory polymer embolic device - European Radiology Experimental
Background We investigated the feasibility of aneurysm sac embolization using a novel self-expanding porous shape memory polymer (SMP) device during endovascular aortic abdominal or thoracic aneurysm repair (EVAR). Methods Retrospective analysis of consecutive patients treated at 2 centers in Germany. Patients were treated from January 2019 to July 2021 with follow-up at 7 days and 3, 6, and 12 months. Aneurysm sacs were implanted with SMP devices immediately following endograft placement during the same procedure. Primary endpoint was technically successful SMP-device deployment into the aneurysm sac outside the endograft. Secondary endpoints were changes in aneurysm volume and associated complications (e.g., endoleaks). Results We included 18 patients (16 males), aged 72 ± 9 years, achieving 100% technical success. Mean preprocedure aortic aneurysm sac volume was 195 ± 117 mL with a perfused aneurysm volume of 97 ± 60 mL. A mean of 24 ± 12 SMP devices per patient were used (range 5–45, corresponding to 6.25–56.25 mL expanded embolic material volume). All evaluable patients exhibited sac regression except 2 patients yet to reach 3-month follow-up. At mean 11 ± 7 months (range 3–24), change in aneurysm volume from baseline was -30 ± 21 mL (p < 0.001). In 8 patients, aneurysm regression was observed despite type 2 endoleaks in 6 and type 1A endoleaks in 2, none of them requiring further intervention to date. No morbidity or mortality related to this treatment occurred. Conclusions SMP devices for aortic aneurysm sac embolization during endovascular repair appear feasible and safe in this small case series. Prospective studies are needed. Key points • Shape memory polymer is a novel, self-expanding, porous, and radiolucent embolic device material. • Aortic aneurysm sacs were treated with polymer devices immediately following endograft placement. • Aortic aneurysm sac regression was observed in all patients with over 3-month follow-up. • Aortic aneurysm sac regression was observed even in the presence of endoleaks.
Authors' #nomogram model showed good discrimination and calibration abilities in the prediction of preoperative acute ischemic #stroke (AIS) in acute type A #aortic disection (ATAAD) patients. (Hongliang Zhao et al.)
🔗 https://link.springer.com/article/10.1007/s00330-023-09691-0
Risk prediction of preoperative acute ischemic stroke in acute type A aortic dissection - European Radiology
Objectives To predict preoperative acute ischemic stroke (AIS) in acute type A aortic dissection (ATAAD). Methods In this multi-center retrospective study, 508 consecutive patients diagnosed as ATAAD between April 2020 and March 2021 were considered for inclusion. The patients were divided into a development cohort and two validation cohorts based on time periods and centers. Clinical data and imaging findings obtained were analyzed. Univariable and multivariable logistic regression analyses were performed to identify predictors associated with preoperative AIS. The performance of resulting nomogram was evaluated in discrimination and calibration on all cohorts. Results A total of 224 patients were in the development cohort, 94 in the temporal validation cohort, and 118 in the geographical validation cohort. Six predictors were identified: age, syncope, D-dimer, moderate to severe aortic valve insufficiency, diameter ratio of true lumen in ascending aorta < 0.33, and common carotid artery dissection. The nomogram established showed good discrimination (area under the receiver operating characteristic curve [AUC], 0.803; 95% CI: 0.742, 0.864) and calibration (Hosmer-Lemeshow test p = 0.300) in the development cohort. External validation showed good discrimination and calibration abilities in both temporal (AUC, 0.778; 95% CI: 0.671, 0.885; Hosmer-Lemeshow test p = 0.161) and geographical cohort (AUC, 0.806; 95% CI: 0.717, 0.895; Hosmer-Lemeshow test p = 0.100). Conclusions A nomogram, based on simple imaging and clinical variables collected on admission, showed good discrimination and calibration abilities in predicting preoperative AIS for ATAAD patients. Key Points • A nomogram based on simple imaging and clinical findings may predict preoperative acute ischemic stroke in patients with acute type A aortic dissection in emergencies. • The nomogram showed good discrimination and calibration abilities in validation cohorts.
#Medical #Cardiology #sflorg
https://www.sflorg.com/2023/03/med03072301.html