Jesseerasmus
- 58 Followers
- 98 Following
- 46 Posts
Director of Virology at HDT Bio. Exploiting RNA viruses for next generation RNA vaccine technologies. Seattle, WA.
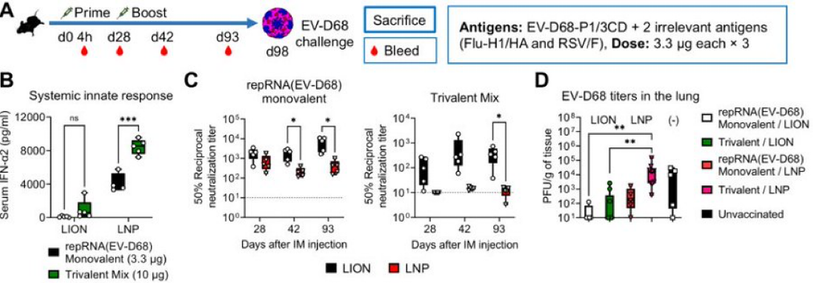
8/ Compared to monovalent EV-D68 vaccine controls, multivalent LNP induced a higher systemic type I IFN response and a reduced neutralizing antibody response. Meanwhile, multivalent LION mitigated the former and maintained the latter, providing superior protection from challenge. Our conclusion? Restricted biodistribution of repRNA is crucial for reducing reactogenicity and enabling adaptive immune responses to multivalent repRNA vaccination. Stay tuned for further developments in our research!
6/ Alongside reactogenicity, we explored whether such systemic responses could affect adaptive immune responses. Despite systemic innate response differences, no variances in binding antibody responses were detected between LNP or LION delivered repRNA vaccines.
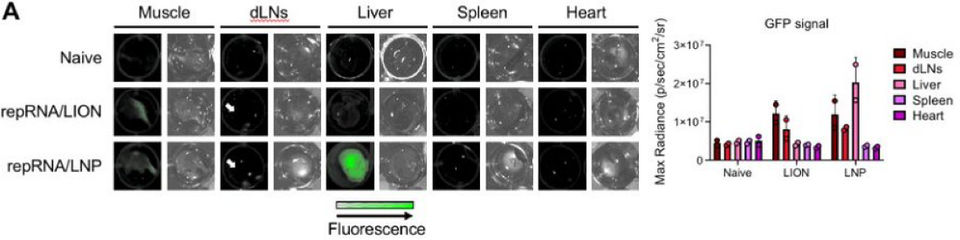
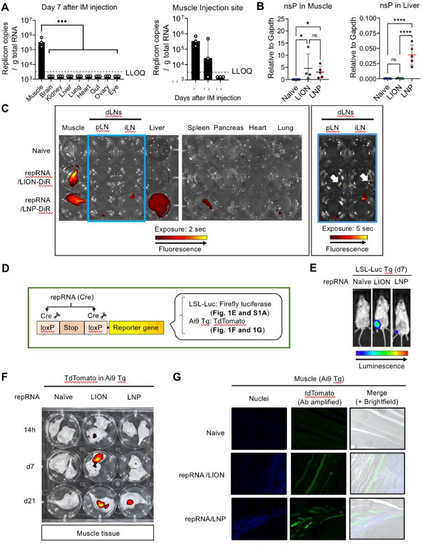
5/ Our data in Mx1/GFP mice suggest the liver is the main contributor to the systemic response to LNP/repRNA.
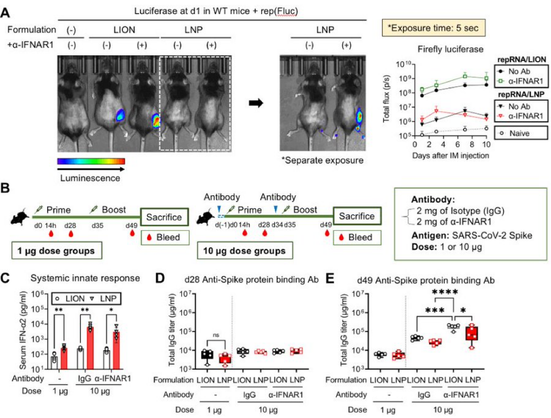
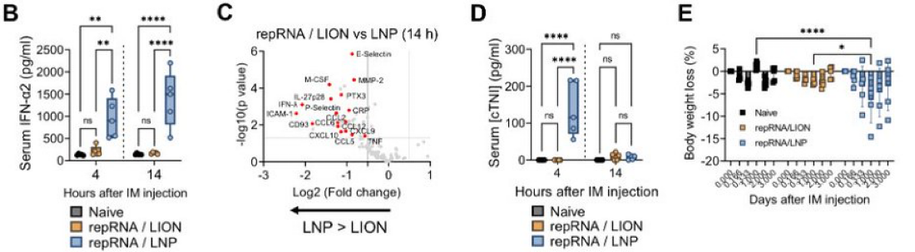
4/ The systemic response was his next focus. He found significant LNP-induced weight loss and high levels of circulating pro-inflammatory cytokines similar to those recently reported by Barmada et al https://twitter.com/anisbarmada/status/1654570291601764354?s=46&t=vbGjOQoWpKGJUKPs7BAxNg. LION, in contrast, caused minimal weight loss and systemic innate response
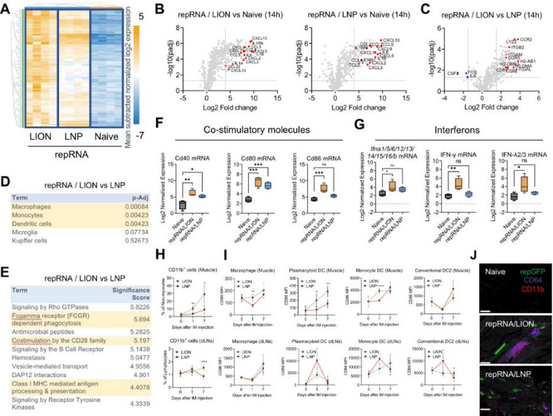
3/ Given the shared delivery site between LION and LNP (muscle), he next evaluated local innate responses in the injection site. He discovered distinct phenotypes, including innate gene regulation and infiltrating immune cells.
2/ As repRNA clinical trial results began to surface, we noticed a difference in dose-dependent reactogenicity. LNP trials reported grade 3 adverse events at lower doses than our findings. With broad biodistribution and systemic innate responses reported for LNP/repRNA, Taishi hypothesized LION might mediate differential biodistribution and systemic response. His research using mice showed that LION stayed primarily at the local injection site, while LNP distributed more widely.
1/ Our team at HDT bio just posted a new paper to Molecular Therapy (https://www.sciencedirect.com/science/article/pii/S1525001623003738). Led by Senior Scientist Taishi Kimura, we investigated the distinct host responses to repRNA delivered by two different formulations: LION and LNP. LION, a cationic nanocarrier, binds RNA at the nanoparticle surface, whereas LNP encapsulates RNA. These different presentations create distinct surface properties and host responses.
Today’s catch from Banner Forest
Thinking about explanations for recent increase in respiratory disease incidence, I’ve had similar thoughts to John’s in this article: https://www.nature.com/articles/d41586-022-03666-9.
Tried doing a quick search for any primary literature to support this hypothesis and came across this review: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8224680/.
Curious to hear if others know of any other support for or against this hypothesis.
This opportunity will provide training in discovery research at a biotechnology company (www.hdt.bio) with multiple products currently in clinical development (including one that achieved EUA)... (3/6)