https://doi.org/doi:10.1136/bmjno-2025-001477
https://pubmed.ncbi.nlm.nih.gov/41877736/
#Gfap
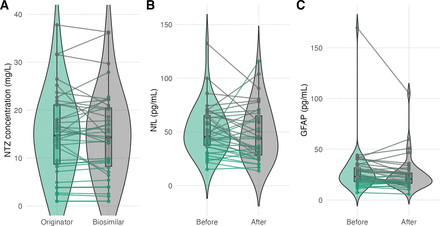
Serum drug levels and JCV assay discrepancies after switching from originator to biosimilar natalizumab
Background In January 2024, all people with multiple sclerosis (pwMS) treated with natalizumab (NTZ) at Oslo University Hospital were switched from originator to biosimilar NTZ. We prospectively evaluated disease activity, safety, serum drug levels, immunogenicity and biomarkers.Methods This observational study included 39 pwMS switching to biosimilar NTZ. Clinical relapses, MRI activity and side effects were recorded. NTZ levels and anti-drug antibodies (ADAb) by in-house assays; anti-John Cunningham virus (JCV) antibodies by Stratify (Biogen) and Immunowell (Sandoz) platforms; leucocytes and serum levels of neurofilament light chain (NfL) and glial fibrillar acidic protein (GFAP) were measured by Mesoscale platform.Results Eleven pwMS (28%) reported new side effects, most commonly fatigue, headache and muscle pain. Mean NTZ levels were 15.1 mg/L before and 14.9 mg/L after switching (difference –0.3, 95% CI –1.4 to 0.8). ADAb was detected in one pwMS, unchanged after switch. The proportion of JCV-positive cases increased from 13% (Stratify) to 52% (Immunowell), leading four pwMS to discontinue NTZ. Leukocytes were stable after switch. Median NfL remained stable (45.3 vs 44.0 pg/mL; median difference –1.3, 95% CI –6.0 to 3.5), whereas GFAP decreased (23.0 vs 20.5 pg/mL; difference –2.5, 95% CI –4.7 to –0.3).Conclusions Switching from originator to biosimilar NTZ was associated with stable disease activity. Drug levels, ADAb, leukocytes and NfL remained similar before and after switching, and GFAP decreased, of uncertain relevance. The marked rise in JCV-positivity on the Immunowell assay underscores the need for harmonisation of anti-JCV antibody testing.